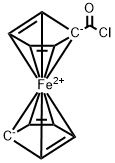
Chlorocarbonyl ferrocene synthesis
- Product Name:Chlorocarbonyl ferrocene
- CAS Number:1293-79-4
- Molecular formula:C11ClFeO
- Molecular Weight:239.42

79-37-8
465 suppliers
$17.67/10gm:
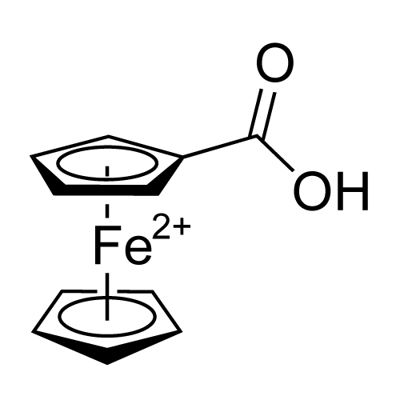
1271-42-7
287 suppliers
$6.00/1g

1293-79-4
58 suppliers
$41.00/1g
Yield:1293-79-4 97%
Reaction Conditions:
in dichloromethane at 0 - 20; for 4 h;Inert atmosphere;
Steps:
3.1 Ferrocenoyl chloride 1
Under a nitrogen atmosphere, to a stirred solution of ferrocene carboxylic acid (1.20 g, 5.2 mmol) in freshly distillated dichloromethane (10 ml), was added dropwise oxalyl chloride (4 ml, 46.8 mmol), at 0 °C.
The resulting mixture was stirred at ambient temp. for 4 h, then the solvent was removed under reduce pressure.
The solution was triturated with hot pentane, then the mixture was filtered, and the filtrate was concentrated under reduced pressure.
The resulting residue was crystallized from pentane to give a red crystalline solid (1.25 g, 97%): mp 134 °C.
1H-NMR (300 MHz, CDCl3): δ 4.36 (s, 5H, C5H5), 4.66 (s, 2H, C5H4), 4.94 (s, 2H, C5H4).
References:
Saied, Nadia Malek;Mejri, Najoua;El Aissi, Radhia;Benoist, Eric;Saidi, Mouldi [European Journal of Medicinal Chemistry,2015,vol. 97,p. 280 - 288]

1271-42-7
287 suppliers
$6.00/1g

1293-79-4
58 suppliers
$41.00/1g
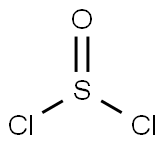
7719-09-7
391 suppliers
$15.00/10g

1271-42-7
287 suppliers
$6.00/1g

1293-79-4
58 suppliers
$41.00/1g
102-54-5
451 suppliers
$5.00/25g
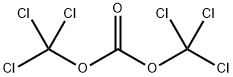
32315-10-9
411 suppliers
$10.00/1g

1293-79-4
58 suppliers
$41.00/1g