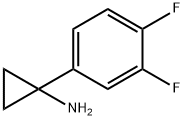
Cyclopropanamine, 1-(3,4-difluorophenyl)- synthesis
- Product Name:Cyclopropanamine, 1-(3,4-difluorophenyl)-
- CAS Number:474709-85-8
- Molecular formula:C9H9F2N
- Molecular Weight:169.17
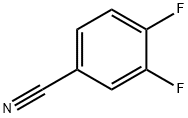
64248-62-0
419 suppliers
$6.00/5g
925-90-6
347 suppliers
$12.00/5ml

474709-85-8
23 suppliers
inquiry
Yield:474709-85-8 56%
Reaction Conditions:
Stage #1: 3,4-Difluorobenzonitrile;ethylmagnesium bromidewith titanium(IV) isopropylate in diethyl ether at -78; for 0.166667 h;
Stage #2: with boron trifluoride diethyl etherate in diethyl ether at -78; for 1 h;
Steps:
14 4.1.14 1-(3,4-Difluorophenyl)cyclopropan-1-amine (18)
To a solution of 3,4-difluorobenzonitrile (278mg, 2mmol) in dry Et2O (10mL) at-78°C was added dropwise Ti(OiPr)4 (0.64mL, 2.2mmol) followed by EtMgBr (1.5mL, 2.2mmol, 3M in Et2O). After 10min, BF3·Et2O (0.5mL, 4mmol) was added and the solution was stirred for 1hat-78°C. The reaction was quenched by addition of 1N HCl (2mL) and diluted with Et2O (10mL). The organic layer was separated, washed with water, brine and dried over MgSO4. The volatiles were evaporated under reduced pressure and the residue was purified by silica gel column chromatogrpahy using hexanes/EtOAc (1:1) to afford 18 (189mg, 56%). 1H NMR (400MHz, Chloroform-d) δ 7.16-7.04 (m, 2H), 7.03-6.98 (m, 1H), 1.91 (s, 2H), 1.16-1.05 (m, 2H), 1.01-0.90 (m, 2H). 13C NMR (101MHz, Chloroform-d) δ 150.1 (dd, J=247.5, 12.8Hz), 148.6 (dd, J=246.2, 12.8Hz), 144.2 (dd, J=5.0, 3.5Hz), 121.2 (dd, J=6.2, 3.4Hz), 116.9 (d, J=17.0Hz), 114.7 (d, J=17.7Hz), 36.2, 18.2. 19F NMR (377MHz, Chloroform-d) δ-139.3 to-139.4 (m),-143.3 to-143.4 (m). HRMS (ESI): m/z [M+H]+ calcd for C9H10F2N: 170.0781, found: 170.0776.
References:
Sari, Ozkan;Boucle, Sebastien;Cox, Bryan D.;Ozturk, Tugba;Russell, Olivia Ollinger;Bassit, Leda;Amblard, Franck;Schinazi, Raymond F. [European Journal of Medicinal Chemistry,2017,vol. 138,p. 407 - 421]