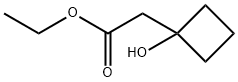
ETHYL 2-(1-HYDROXYCYCLOBUTYL)ACETATE synthesis
- Product Name:ETHYL 2-(1-HYDROXYCYCLOBUTYL)ACETATE
- CAS Number:27784-32-3
- Molecular formula:C8H14O3
- Molecular Weight:158.2
Yield:27784-32-3 54%
Reaction Conditions:
Stage #1: ethyl bromoacetatewith chloro-trimethyl-silane;zinc in diethyl ether at 20; for 2 h;Reflux;Reformatsky reaction;
Stage #2: cyclobutanone in diethyl ether at 19 - 20; for 1 h;Reformatsky reaction;
Stage #3: with ammonia in diethyl ether;water;Cooling with ice;
Steps:
5.1.38. Ethyl 1-hydroxycyclobutylacetate (50)
Trimethylchlorosilane (1.14 mL, 8.92 mmol) was added from a syringe to a suspension of zinc powder (7.97 g, 0.122 mol) in Et2O (200 mL). The mixture was stirred for 15 min at room temperature and then heated to reflux. The heating was stopped, and ethyl bromoacetate (10.3 mL, 92.9 mmol) was added at such a rate that the ether boiled gently. After being heated to reflux for 1 h, the mixture was stirred for 1 h at room temperature. A solution of 48 (6.00 g, 75.9 mmol) in Et2O (30 mL) was added while the temperature of the mixture was maintained at 19-20 °C by intermittent cooling. After being stirred for 1 h at room temperature, the mixture was poured into iced 25% ammonia (400 mL). The aqueous phase was extracted with ether, the combined phases were dried on K2CO3 and evaporated, and 50 was obtained as a colorless oil (6.50 g, 54%). 1H NMR (400 MHz, CDCl3) δ 1.29 (3H, t, J = 7.3 Hz), 1.47-1.64 (1H, m), 1.76-1.87 (1H, m), 1.93-2.06 (2H, m), 2.12-2.22 (2H, m), 2.67 (2H, s), 3.70 (1H, s), 4.19 (2H, q, J = 7.3 Hz). HRMS (CI+) for C8H15O3 [M+H]+: calcd, 159.1021; found, 159.0994.
References:
Seto, Shigeki;Yumoto, Kazuhiko;Okada, Kyoko;Asahina, Yoshikazu;Iwane, Aya;Iwago, Maki;Terasawa, Reiko;Shreder, Kevin R.;Murakami, Koji;Kohno, Yasushi [Bioorganic and Medicinal Chemistry,2012,vol. 20,# 3,p. 1188 - 1200] Location in patent:experimental part
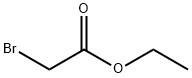
105-36-2
346 suppliers
$5.00/5G

27784-32-3
18 suppliers
inquiry
