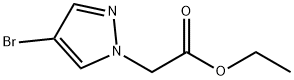
Ethyl 2-(4-broMo-1H-pyrazol-1-yl)acetate synthesis
- Product Name:Ethyl 2-(4-broMo-1H-pyrazol-1-yl)acetate
- CAS Number:82231-58-1
- Molecular formula:C7H9BrN2O2
- Molecular Weight:233.06
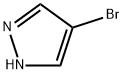
2075-45-8
398 suppliers
$5.00/5g
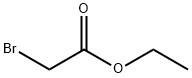
105-36-2
346 suppliers
$5.00/5G

82231-58-1
50 suppliers
$63.00/100mg
Yield:82231-58-1 83%
Reaction Conditions:
Stage #1: 4-bromo-1H-pyrazolewith sodium hydride in N,N-dimethyl-formamide;mineral oil at 0; for 1 h;
Stage #2: ethyl bromoacetatewith potassium iodide in N,N-dimethyl-formamide;mineral oil at 0; for 8 h;Heating;
Steps:
9 6.9 Ethyl 2-(4-bromo-1H-pyrazol-1-yl)acetate (24)
To an ice-cooled solution of 4-bromo-1H-pyrazole (1.00 g, 6.80 mmol) in 10 mL of DMF was added portionwise sodium hydride (327 mg, 8.16 mmol, 60% in oil). The resulting mixture was stirred for additional 1 h. Then to the mixture was added ethyl 2-bromoacetate (0.83 mL, 7.48 mmol) and KI (226 mg, 1.36 mmol). The resulting mixture was gradually heated to 80 °C and stirred for 8 h. After cooled to room temperature, the reaction was quenched with 20 mL of water. The mixture was extracted with EA (2 × 75 mL). The combined organic layer was washed with water (3 × 15 mL), brine (40 mL), dried over anhydrous Na2SO4 and concentrated under vacuum. The crude product was purified by flash column chromatography (0-10% EA/PE gradient) to afford 24 as clear colorless oil in 83% yield. 1H NMR (300 MHz, CDCl3) δ 7.51 (s, 2H), 4.87 (s, 2H), 4.24 (q, J = 6.9 Hz, 2H), 1.29 (t, J = 6.9 Hz, 3H).
References:
Zhang, Dengyou;Zhang, Xiaowei;Ai, Jing;Zhai, Yun;Liang, Zhongjie;Wang, Ying;Chen, Yi;Li, Chunpu;Zhao, Fei;Jiang, Hualiang;Geng, Meiyu;Luo, Cheng;Liu, Hong [Bioorganic and Medicinal Chemistry,2013,vol. 21,# 21,p. 6804 - 6820]

2075-45-8
398 suppliers
$5.00/5g
105-39-5
382 suppliers
$10.00/5g

82231-58-1
50 suppliers
$63.00/100mg