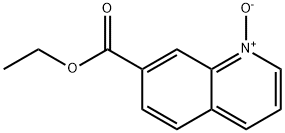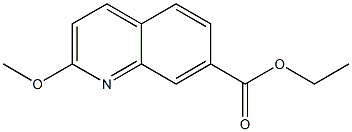
Ethyl 2-methoxyquinoline-7-carboxylate synthesis
- Product Name:Ethyl 2-methoxyquinoline-7-carboxylate
- CAS Number:1374258-39-5
- Molecular formula:C13H13NO3
- Molecular Weight:231.2472
Yield:1374258-39-5 81%
Reaction Conditions:
with triethylamine;p-toluenesulfonyl chloride at 0 - 20;
Steps:
19.2
To a 0° C. solution of 7-(ethoxycarbonyl)quinoline 1-oxide (150 mg, 0.69 mmol) and toluene-4-sulphonyl chloride (171 mg, 0.89 mmol) in methanol (5 mL) was added triethylamine (0.19 mL, 1.4 mmol). The reaction was stirred at room temperature overnight. LCMS showed the reaction was incomplete. Additional triethylamine (0.05 mL) was added and the reaction was stirred for another 4 hours. The reaction was concentrated and the residue partitioned between ethyl acetate and saturated aqueous sodium carbonate. The layers were separated and the aqueous was extracted two more times with ethyl acetate. The combined organics were washed with brine, dried over sodium sulfate, filtered, and concentrated. Purification by flash column chromatography (0-40% ethyl acetate/heptanes) gave the title compound (130 mg, 81%) as a pale yellow solid. +ESI (M+H) 232.2; 1H NMR (400 MHz, CDCl3, δ): 8.49-8.60 (m, 1H), 7.95-8.05 (m, 2 H), 7.75 (d, J=8.19 Hz, 1H), 6.98 (d, J=8.78 Hz, 1H), 4.43 (q, J=7.22 Hz, 2H), 4.08 (s, 3H), 1.43 (t, J=7.12 Hz, 3H).
References:
US2012/108619,2012,A1 Location in patent:Page/Page column 36
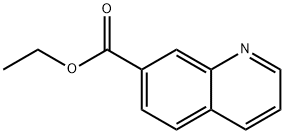
104294-00-0
24 suppliers
inquiry

1374258-39-5
4 suppliers
inquiry