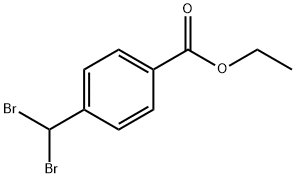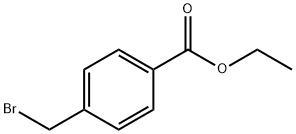
Ethyl 4-(bromomethyl)benzoate synthesis
- Product Name:Ethyl 4-(bromomethyl)benzoate
- CAS Number:26496-94-6
- Molecular formula:C10H11BrO2
- Molecular Weight:243.1

64-17-5
705 suppliers
$10.00/50g

6232-88-8
316 suppliers
$10.00/1g

26496-94-6
107 suppliers
$10.00/5g
Yield:26496-94-6 75%
Reaction Conditions:
with dmap;dicyclohexyl-carbodiimide in dichloromethane at 0; for 12 h;Inert atmosphere;
Steps:
Syntheses of compounds 6e-6j
General procedure: General Method H: To a solution of the acid (1.0 equiv.) in DCM, DCC (1.5 equiv.) was added and followed byalcohol (1.0 equiv.). Catalytic amount of DMAP was then added to the mixture. The reaction mixture was stirredovernight under N2. Water (5.0 mL) was added and the mixture was extracted with DCM. The organic phase waswashed with brine, dried over Na2SO4, filtered and concentrated. The residue was purified by silica gelchromatography (EtOA/hexane) to afford pure products.
References:
Liu, Hao;Chen, Li;Zhou, Fei;Zhang, Yun-Xiao;Xu, Ji;Xu, Meng;Bai, Su-Ping [Bioorganic and Medicinal Chemistry,2019,vol. 27,# 14,p. 3089 - 3096] Location in patent:supporting information

94-08-6
216 suppliers
$6.00/5g

26496-94-6
107 suppliers
$10.00/5g

64-17-5
705 suppliers
$10.00/50g
52780-16-2
15 suppliers
$111.00/5mg

26496-94-6
107 suppliers
$10.00/5g

99-94-5
569 suppliers
$9.00/1g

26496-94-6
107 suppliers
$10.00/5g