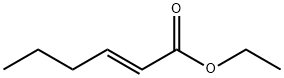
Ethyl (E)-hex-2-enoate synthesis
- Product Name:Ethyl (E)-hex-2-enoate
- CAS Number:27829-72-7
- Molecular formula:C8H14O2
- Molecular Weight:142.2

123-72-8
402 suppliers
$12.32/25ML
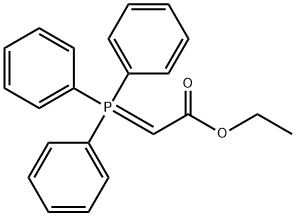
1099-45-2
362 suppliers
$6.00/5g

27829-72-7
183 suppliers
$16.00/10g
Yield:27829-72-7 72%
Reaction Conditions:
in dichloromethane at 20; for 47 h;Inert atmosphere;Wittig Olefination;
Steps:
Synthesis and data for (E)-ethyl hex-2-enoate (6):
To a stirred solution of butanal(0.213 g, 2.95 mmol) in CH2Cl2 (15 mL) under an atmosphere of nitrogen wasadded (carbethoxymethylene)triphenylphosphorane (1.24 g, 3.50 mmol) andthe resulting mixture stirred for 47 h. The solvent was removed in vacuo,without application of heat, and pentane (30 mL) was added to the residue,which was then stirred for 30 min. The resulting mixture was filtered, thesolvent removed in vacuo, without application of heat, and the crude productwas purified using flash chromatography (95:5 pentane/Et2O) to give the ester6 (0.30 g, 72%) as a pale yellow oil. dH (400 MHz, CDCl3): 0.93 (3H, t, J = 7.5 Hz,6-CH3), 1.28 (3H, t, J = 7.0 Hz, 8-CH3), 1.44-1.53 (2H, m, 5-CH2), 2.14-2.20 (2H,m, 4-CH2), 4.18 (2H, q, J = 7.0 Hz, 7-CH2), 5.81 (1H, dt, J = 1.5, 15.9 Hz, 2-H), 6.96(1H, dt, J = 7.1, 15.2 Hz, 3-H); dC (100 MHz, CDCl3): 13.8 (C-6), 14.4 (C-8), 21.4(C-5), 34.3 (C-4), 60.2 (C-7), 121.5 (C-2), 149.3 (C-3), 166.9 (C-1).
References:
Duhamel, Nina;Piano, Federico;Davidson, Samuel J.;Larcher, Roberto;Fedrizzi, Bruno;Barker, David [Tetrahedron Letters,2015,vol. 56,# 13,p. 1728 - 1731]

6728-26-3
327 suppliers
$19.00/25g

64-17-5
707 suppliers
$10.00/50g

27829-72-7
183 suppliers
$16.00/10g
867-13-0
465 suppliers
$10.00/1g

123-72-8
402 suppliers
$12.32/25ML

27829-72-7
183 suppliers
$16.00/10g
2396-84-1
155 suppliers
$11.00/10g
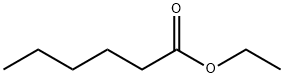
123-66-0
338 suppliers
$10.00/5g

27829-72-7
183 suppliers
$16.00/10g

26553-46-8
21 suppliers
inquiry

64187-83-3
5 suppliers
inquiry

123-72-8
402 suppliers
$12.32/25ML

27829-72-7
183 suppliers
$16.00/10g
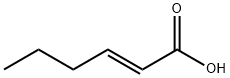
13419-69-7
249 suppliers
$10.00/1g