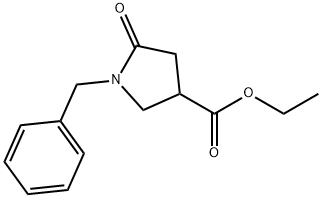
ETHYL1-BENZYL-5-OXOPYRROLIDINE-3-CARBOXYLATE synthesis
- Product Name:ETHYL1-BENZYL-5-OXOPYRROLIDINE-3-CARBOXYLATE
- CAS Number:5733-87-9
- Molecular formula:C14H17NO3
- Molecular Weight:247.29
Yield:-
Reaction Conditions:
in ethanol;Reflux;
Steps:
23.(1-Benzylpyrrolidin-3-yl)methanol (31).
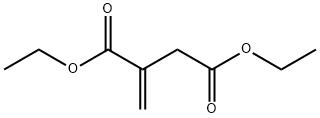
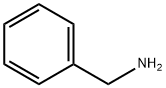
A solution of diethyl itaconate (150 g, 807 mmol) and benzylamine (29) (86.4 g, 807 mmol) in ethanol (90 mL) was refluxed for overnight. The mixture was cooled to room temperature and concentrated in vacuo. The residue was purified by silica gel column chromatography (hexane : ethyl acetate = 80 : 20 - 50 : 50) to afford oil (190 g). A part of the oil (42 g) was dilluted with tetrahydrofuran (200 mL). To the solution was added dropwise a solution of lithium aluminum hydride (10% in tetrahydrofuran, ca. 2.4 M, 100 mL, 240 mmol) at 0 °C over 2 hours. The mixture was stirred at 50 °C for overnight. It was cooled to 0 °C and added sodium sulfate decahydrate then filtered through celite. The filtrate was concentrated in vacuo to give the title compound (35 g, 183 mmol, quant.) as color less oil.1H-NMR (CDCl3): δ 1.641.75 (m, 1H), 1.942.06 (m, 1H), 2.282.37 (m, 3H), 2.472.53 (m, 1H), 2.612.65 (m, 1H), 2.772.85 (m, 1H), 3.493.54 (m, 1H), 3.59 (s, 2H), 3.643.69 (m, 1H), 7.217.33 (m, 5H).
References:
Nakamura, Shinji;Kamaura, Masahiro;Akao, Yuichiro;Nakamura, Natsuko;Mizukami, Atsushi;Goto, Akihiko;Furuyama, Naoki;Cho, Nobuo;Kasai, Shizuo [Bioorganic and Medicinal Chemistry,2022,vol. 54,art. no. 116553] Location in patent:supporting information