
H-BETA,BETA-DIMETHYL-D-CYS(PMEOBZL)-OH synthesis
- Product Name:H-BETA,BETA-DIMETHYL-D-CYS(PMEOBZL)-OH
- CAS Number:53599-14-7
- Molecular formula:C13H19NO3S
- Molecular Weight:269.36
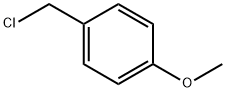
824-94-2
442 suppliers
$24.19/5G

53599-14-7
13 suppliers
$40.00/250MG
Yield:53599-14-7 33%
Reaction Conditions:
with trifluoroacetic acid in methanol;dichloromethane;water;
Steps:
47.a 47a.
47a. (2S)-2-Amino-3-[(4-methoxyphenyl)methylthio]-3-methylbutanoic acid A suspension of (2S)-amino-3-methyl-3-sulfanylbutanoic (11.5 g, 77.2 mmol) in CH2Cl2 (60 mL) was treated with trifluoroacetic acid (13.7 mL) and stirred to dissolve at room temperature. The mixture was then cooled to -10° C. under nitrogen. A solution of 4-methoxybenzyl chloride (10.5 mL, 77.25mmol) in CH2Cl2 (90 mL) was added dropwise through an additional funnel over a period of 1.5 hours. Stirring was continued for 1.5 hours at room temperature. Methanol (10 mL) was added to dissolve the precipitate. The crude reaction was concentrated in vacuo. The residue was dissolved in CH2Cl2 (40 mL) and extracted with water (7*50 mL). The combined aqueous extracts were frozen and lyophilized. The residue was dissolved in methanol/water (1:3, 200 mL) and brought to pH 6-7 with sodium bicarbonate. The white solid was isolated by filtration, rinsed with MeOH/water (1:3), and dried to give the title compound (6.92 g, 33%). 1H NMR (300 MHz, CD3OD) δ 7.28 (d, J=7.0 Hz, 2H), 6.85 (d, J=8.4 Hz, 2H), 3.79-3.76 (m, 5H), 3.51 (s, 1H), 1.63 (s, 3H), 1.33 (s, 3H).
References:
US6297260,2001,B1