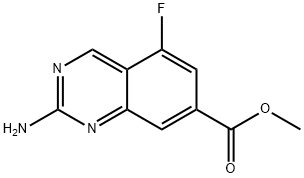
METHYL 2-AMINO-5-FLUOROQUINAZOLINE-7-CARBOXYLATE synthesis
- Product Name:METHYL 2-AMINO-5-FLUOROQUINAZOLINE-7-CARBOXYLATE
- CAS Number:1422188-61-1
- Molecular formula:C10H8FN3O2
- Molecular Weight:221.19

67-56-1
739 suppliers
$9.00/25ml
201230-82-2
1 suppliers
inquiry
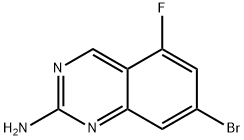
1379341-83-9
23 suppliers
$220.00/100mg

1422188-61-1
7 suppliers
inquiry
Yield:1422188-61-1 59%
Reaction Conditions:
with 1,3-bis(dicyclohexylphosphino)propane bis(tetrafluoroborate);palladium diacetate;potassium carbonate in N,N-dimethyl-formamide at 100; for 6 h;Inert atmosphere;
Steps:
8.7
[002961 step 7: 7-Bromo-5-fluoro-quinazolin-2-amine (8.02 g, 33.1 mmol, 1.00 equiv), Pd(OAc)2 (750 mg, 3.34 mmol, 0.10 equiv), l,3-bis(dicyclohexylphosphino)propane bis(tetrafluoroborate) (2.04 g, 3.22 mmol, 0.10 equiv) and K2C03 (9.27 g, 66.4 mmol, 2.00 equiv) were suspended together in DMF (100 ml) under nitrogen and treated with anhydrous MeOH (13.5 mL, 333 mmol, 10 equiv). The flask was thoroughly flushed with carbon monoxide, and then heated to 100 °C with a continuous flow of carbon monoxide, which was switched to a static positive pressure on reaching temperature. After 6 h the reaction was cooled. The mixture was diluted with water (200 mL) and extracted twice into EtOAc. The combined organic phases were washed with water and brine, dried (Na2S04), filtered and concentrated in vacuo to afford a crude orange solid (6.76 g) which was absorbed onto Si02 and purified by automated Si02 chromatography eluting with an EtOAc/heptane gradient (0 to 100% EtOAc) to afford 4.30 g (59%) of methyl 2-amino-5- fluoroquinazoline-7-carboxylate as a pale orange solid: 1H NMR (400 MHz, CDC13) δ 9.35 (s, 1H), 8.07 (s, 1H), 7.48 (dd, J= 10.0, 1.0 Hz, 1H), 5.35 (br s, 2H), 3.98 (s, 3H); LCMS: MH+ 222.2
References:
WO2013/20062,2013,A1 Location in patent:Paragraph 00296
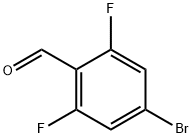
537013-51-7
220 suppliers
$10.00/1g

1422188-61-1
7 suppliers
inquiry