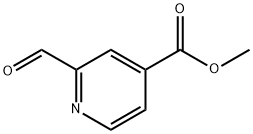
Methyl 2-forMylisonicotinate synthesis
- Product Name:Methyl 2-forMylisonicotinate
- CAS Number:125104-34-9
- Molecular formula:C8H7NO3
- Molecular Weight:165.15

58481-17-7
78 suppliers
$155.00/100mg

125104-34-9
72 suppliers
$25.00/100mg
Yield:125104-34-9 85%
Reaction Conditions:
Stage #1:methyl 2-(hydroxymethyl)pyridine-4-carboxylate with Dess-Martin periodane in dichloromethane at 20; for 2.5 h;
Stage #2: with sodium hydrogencarbonate in dichloromethane;water at 20; for 0.25 h;
Steps:
39.1
Methyl 2-(hydroxymethyl)isonicotinate (8.36 g, 50 mmol) (from reference compound 38, step 1) was dissolved in dichloromethane (150 mL). Dess-Martin periodinane (25 g, 58.94 mmol) was added and the mixture stirred at room temperature for 2 h 30 min. Sodium sulfothioate (59.3 g, 375.00 mmol) was dissolved in satd NaHCO3 and added to the reaction mixture. The suspension was vigorously stirred at room temperature for 15 min, DCM was added and the phases were separated. The aqueous phase was extracted with DCM twice and the combined organic layers were dried over MgSO4 and evaporated. The residue was purified by automated flash chromatography on a Biotage KP-SIL 340 g column. Gradient heptanes/EtOAc 80:20 to 65:35 over 5 CV was used as mobile phase. Methyl 2-formylisonicotinate (7 g, 85%) was isolated as an off-white solid. 1H NMR (400 MHz, cdcl3) δ 4.00 (s, 3H), 8.09 (dd, 1H), 8.49 (s, 1H), 8.95 (d, 1H), 10.15 (s, 1H). MS m/z 165 (M)+
References:
AstraZeneca AB US2010/261755, 2010, A1 Location in patent:Page/Page column 35
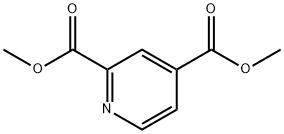
25658-36-0
64 suppliers
$45.00/50mg

125104-34-9
72 suppliers
$25.00/100mg
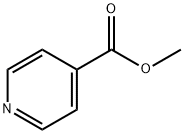
2459-09-8
287 suppliers
$7.00/10g

125104-34-9
72 suppliers
$25.00/100mg