
Methyl 4-(4-Boc-1-piperazinyl)-3-chlorobenzoate synthesis
- Product Name:Methyl 4-(4-Boc-1-piperazinyl)-3-chlorobenzoate
- CAS Number:906559-46-4
- Molecular formula:C17H23ClN2O4
- Molecular Weight:354.83
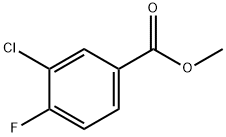
234082-35-0
92 suppliers
$16.00/1g
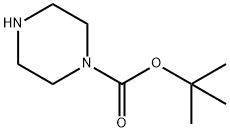
57260-71-6
733 suppliers
$5.00/5g

906559-46-4
25 suppliers
$60.00/100mg
Yield:906559-46-4 74%
Reaction Conditions:
with potassium carbonate in dimethyl sulfoxide at 100; for 10 h;Sealed tube;
Steps:
2.2 Step-2
Methyl3-chloro-4-fluoro-benzoate (1.5 g, 7.9 mmol) was taken in a sealed tube, piperazine-1-carboxylic acid tert-butyl ester (1.48 g, 7.9 mmol) followed by K2C03 (3.29 g, 23 mmol)and DMSO (15 mL) were added and stirred at 100 °C for 10 h (TLC indicated completeconsumption of starting material). The reaction mixture was poured into ice-cold water (150mL) and extracted with EtOAc (2 x 100 mL). The combined organic extracts were washedwith brine (2 x 75 mL), dried over Na2S04 and concentrated under reduced pressure to givethe crude residue which was purified by column chromatography (100-200 silica gel, 30 g,10% EtOAc-hexane) to afford tert-butyl4-(2-chloro-4-methoxycarbonyl-phenyl)piperazinel-carboxylate (2.1 g, 74%) as a white solid LCMS: m/z: 355.4 [M+Ht.
References:
WO2018/125961,2018,A1 Location in patent:Page/Page column 30; 31
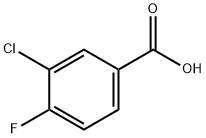
403-16-7
273 suppliers
$6.00/5g

906559-46-4
25 suppliers
$60.00/100mg