
Methyl 4-AMino-5-chloro-2-Methoxy-3-nitrobenzoate synthesis
- Product Name:Methyl 4-AMino-5-chloro-2-Methoxy-3-nitrobenzoate
- CAS Number:457947-61-4
- Molecular formula:C9H9ClN2O5
- Molecular Weight:260.63
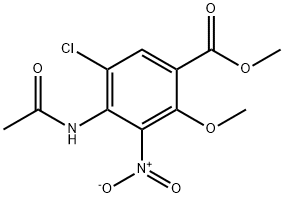
126463-85-2
9 suppliers
inquiry

457947-61-4
14 suppliers
inquiry
Yield:457947-61-4 99%
Reaction Conditions:
Stage #1: 4-acetylamino-5-chloro-2-methoxy-3-nitro-benzoic acid methyl esterwith sulfuric acid in methanol; for 7 h;Reflux;
Stage #2: with sodium hydrogencarbonate in water;
Steps:
I.I-XVIII.I-FD
General Procedure I-FDTo a solution of compound I-XVIIIa (15.0 g, 49.7 mmol) in 100 mL of MeOH was added 6 mL of conc. H2SO4. The solution was heated to reflux for 7 hrs. Subsequently, the solution was concentrated under reduced pressure. The residue was diluted with water (30 mL), and then neutralized by addition of saturated aq. NaHCO3, the solution was extracted with EtOAc (30 mL×3), the organic layer was washed with water and brine, dried over anhydrous Na2SO4, and concentrated in vacuo to give compound I-XVIIIb (12.8 g, yield 99%) as yellow solid.
References:
US2011/152246,2011,A1 Location in patent:Page/Page column 193

4093-31-6
224 suppliers
$8.00/5g

457947-61-4
14 suppliers
inquiry
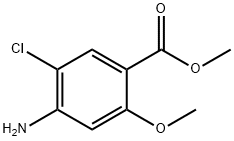
20896-27-9
65 suppliers
$45.00/50mg

457947-61-4
14 suppliers
inquiry
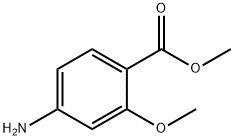
27492-84-8
310 suppliers
$6.00/5g

457947-61-4
14 suppliers
inquiry