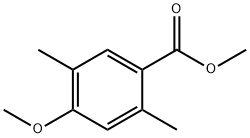
Methyl 4-Methoxy-2,5-diMethylbenzoate synthesis
- Product Name:Methyl 4-Methoxy-2,5-diMethylbenzoate
- CAS Number:58106-28-8
- Molecular formula:C11H14O3
- Molecular Weight:194.23
Yield:58106-28-8 23%
Reaction Conditions:
with iodine;potassium hydroxide at 0; for 1.5 h;
Steps:
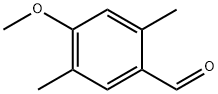
Synthesis of methyl 2,5-dimethyl-p-anisate (26)
Similar to a reported procedure [39], 2,5-dimethyl-p-anisaldehyde (25, 1 g, 6.09 mmol, 1 equiv) was dissolved in MeOH (60 mL) and the solution was cooled to 0 °C. Solutions of KOH (1.045 g, 15.89 mmol, 2.6 equiv, in 20 mL MeOH) and I2 (2.01 g, 7.92 mmol, 1.3 equiv, in 10 mL MeOH) were added and the mixture was stirred for 90 min at 0 °C. The reaction was diluted with EtOAc, washed three times with saturated aqueous Na2S2O3 solution and subsequently with brine. The organic layer was dried over MgSO4 and the solvent was removed under reduced pressure. The crude product was purified via column chromatography (cyclohexane/ethyl acetate 10:1) on silica gel and the pure product was obtained as a colourless solid (277 mg, 1.43 mmol, 23%). TLC (silica, cyclohexane/ethyl acetate 3:1): Rf = 0.67. 1H NMR (500 MHz, CDCl3, 298 K) δ (ppm) 7.75 (s, 1H, CH), 6.64 (s, 1H, CH), 3.86 (s, 3H, CH3), 3.85 (s, 3H, CH3), 2.60 (s, 3H, CH3), 2.18 (s, 3H, CH3); 13C NMR (125 MHz, CDCl3, 298 K) δ (ppm) 167.9 (Cq), 160.6 (Cq), 140.8 (Cq), 133.4 (CH), 123.9 (Cq), 120.9 (Cq), 112.9 (CH), 55.5 (CH3), 51.6 (CH3), 22.1 (CH3), 15.8 (CH3).
References:
Rinkel, Jan;Babczyk, Alexander;Wang, Tao;Stadler, Marc;Dickschat, Jeroen S. [Beilstein Journal of Organic Chemistry,2018,vol. 14,p. 2974 - 2990]