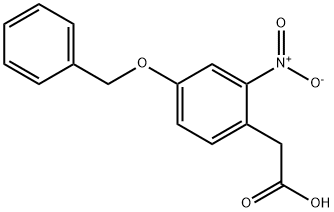
MFCD08142191 synthesis
- Product Name:MFCD08142191
- CAS Number:6860-79-3
- Molecular formula:C15H13NO5
- Molecular Weight:287.27
![Propanedioic acid, 2-[2-nitro-4-(phenylmethoxy)phenyl]-, 1,3-diethyl ester](/CAS/20210305/GIF/112089-51-7.gif)
112089-51-7
0 suppliers
inquiry

6860-79-3
11 suppliers
inquiry
Yield:6860-79-3 95%
Reaction Conditions:
Stage #1: diethyl (4-benzyloxy-2-nitrophenyl)malonatewith sodium hydroxide;water in ethanol; for 3 h;Heating / reflux;
Stage #2: with hydrogenchloride;water in tetrahydrofuran at 0; pH=1;Heating / reflux;
Steps:
1 Preparation of 2-(2-Amino-4-hydroxyphenyl)ethyl Bromide and 2-(2-Amino-4-hydroxyphenyl)ethyl Chloride
To a round-bottom flask containing diethyl 2-(4-benzyloxy-2-nitrophenyl) malonate (4c) (2.00 g, 5.17 mmol) was added EtOH (45 mL) and 10% NaOH (60 mL) producing a dark brown solution. The solution was refluxed for 3 h and checked by TLC (CHCl3), at which time the solution was slightly yellowish and clear. The EtOH was removed under reduced pressure to form a yellow suspension and sufficient THF (30 mL) was added to produce a clear yellow solution which was placed in an ice bath and stirred. 6M HCl (30 mL) was slowly added to the solution to reach pH 1. The light orange solution was refluxed for another hour, at which time two layers formed. The top THF layer was collected, and the aqueous solution was extracted with CHCl3 (100 mL). The organic layers were combined and dried with anhydrous sodium sulfate. The solution was then filtered, and the filtrate was concentrated to produce 4-benzyloxy-2-nitrophenylacetic acid as an orange solid. The solid was then placed on a high vacuum and dried (1.41 g, 4.91 mmol, 95%). Mp 145-152C; TLC (CHCl3) Rf=0.15; 1H-NMR (CDCl3, 300 MHz) 7.76 (d, 2.7, 1H), 7.41 (m, 5H), 7.26 (d, 8.7, 1H), 7.21 (dd, 2.7, 8.7, 1H), 5.13 (s, 2H), 4.00 (s, 2H); IR (CHCl3 cast) 3400-2600 (br), 3100, 1698, 1601, 1526, 1523, 1453, 1376, 1349, 1240, 1180, 1022, 848, 815, 760, 712; EI-MS m/z (relative intensity) 287 (M+, 10). Accurate mass (EI-MS) for C15H13NO5: calcd. 287.0794, obsd. 287.0799.
References:
US6660742,2003,B2 Location in patent:Page column 27
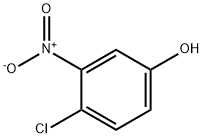
610-78-6
150 suppliers
$7.00/1g

6860-79-3
11 suppliers
inquiry
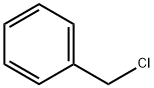
100-44-7
654 suppliers
$13.50/250G

6860-79-3
11 suppliers
inquiry
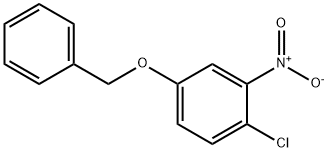
79035-13-5
16 suppliers
inquiry

6860-79-3
11 suppliers
inquiry

