
N-ACRYLOXYSUCCINIMIDE synthesis
- Product Name:N-ACRYLOXYSUCCINIMIDE
- CAS Number:38862-24-7
- Molecular formula:C7H7NO4
- Molecular Weight:169.13
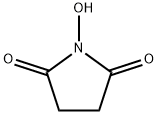
6066-82-6
738 suppliers
$5.00/10g
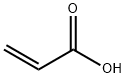
79-10-7
685 suppliers
$20.16/100 mL

2524-64-3
386 suppliers
$14.00/5g

38862-24-7
158 suppliers
$8.00/250mg
Yield:-
Reaction Conditions:
in dichloromethane;di-isopropyl ether;water;acetone
Steps:
11 EXAMPLE 11
EXAMPLE 11 0.72 g (0.010 mol) of acrylic acid was initially introduced into 30 ml of acetone and 1.27 g (0.011 mol) of solid N-hydroxysuccinimide were added. The resulting solution was treated with 3.36 g (0.040 mol) of solid sodium hydrogencarbonate and the white suspension thus obtained was stirred at room temperature. A solution of 3.22 g (0.012 mol) of diphenyl chlorophosphate in 10 ml of acetone was then added dropwise and the reaction mixture was stirred at room temperature for 24 hours and at 50° C. for a further 3 hours to complete the reaction, the thickening suspension being diluted with 25 ml of acetone. The diluent was then removed in vacuo and the residue was taken up in 60 ml of dichloromethane. Undissolved solid was filtered and washed twice with 10 ml each of dichloromethane. The combined dichloromethane phases were washed with 40 ml of water and, after phase separation, evaporated in vacuo. 1.53 g of solid crude product were obtained, which was washed by stirring with 15 ml of diisopropyl ether for 3 hours. The white solid which remained was filtered off, washed twice with 3 ml of diisopropyl ether each time and dried in vacuo. 1.1 g (65%) of acrylic acid succinimidyl ester were obtained. M.p.: 63°-68° C.
References:
DSM Chemie Linz GmbH US5734064, 1998, A

6066-82-6
738 suppliers
$5.00/10g

814-68-6
380 suppliers
$21.21/5gm:

38862-24-7
158 suppliers
$8.00/250mg

6066-82-6
738 suppliers
$5.00/10g

79-10-7
685 suppliers
$20.16/100 mL

38862-24-7
158 suppliers
$8.00/250mg
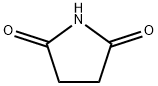
123-56-8
489 suppliers
$6.00/25g

814-68-6
380 suppliers
$21.21/5gm:

38862-24-7
158 suppliers
$8.00/250mg