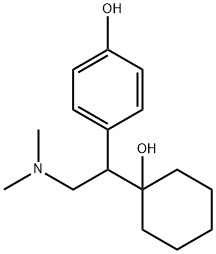
O-Desmethylvenlafaxine synthesis
- Product Name:O-Desmethylvenlafaxine
- CAS Number:93413-62-8
- Molecular formula:C16H25NO2
- Molecular Weight:263.38

93413-69-5
247 suppliers
$10.00/1mg

93413-62-8
230 suppliers
$5.00/5mg
Yield:93413-62-8 98%
Reaction Conditions:
with potassium hydroxide in ethylene glycol at 195 - 200; for 24 h;Inert atmosphere;
Steps:
1 Example 1:
To a clean flask , 250g of 1- [2 -(Dimethyl amino- l-(4-methoxyphenyl) ethyl cyclohexanol, lit of ethylene glycol, 100 g of polyethylene glycol-400 and 350 g of Potassium hydroxide are added, under nitrogen atmosphere. Reaction mixture is then heated under nitrogen, by raising the temperature to about 195-200 °C over a period of about 3-4 h. The same temperature is then, continuously maintained for a period of 24 h. The temperature of the reaction mixture is loweredto a temperature of about 50°C and diluted with water and extracted with lit of toluene. Toluene is concentrated in order to obtain unreacted l-[2-(Dimethyl amino- l-(4-methoxyphenyl) ethyl cyclohexanolof about 95g to about TOOg. Water layer is then acidified to pH 4 to getl-[2- (Dimethylamino)-l-(4-hydroxyphenyl)ethyl]-cyclohexanol.On drying the mixture at a temperature ranging from about 75 °C to about 80°C for 8 hours, l-[2-(Dimethylamino)-l-(4- hydroxyphenyl)ethyl]-cyclohexanol, is obtained (125-130g) with a purity of 99.25% measured by HPLC). Yield percentage based on recovery of unreacted l-[2-(Dimethyl amino-l-(4- methoxyphenyl) ethyl cyclohexanol is 98%.
References:
R L FINECHEM PRIVATE LIMITED;A RAMAKRISHNA, Ramesha;S MARAVANAHALLI, Siddegowda;MAHIL, Santosh WO2018/146529, 2018, A1 Location in patent:Page/Page column 10
![1-[2-Amino-1-(4-benzyloxyphenyl)-ethyl]-cyclohexanol](/CAS/GIF/93413-61-7.gif)
93413-61-7
55 suppliers
$160.00/50mg

93413-62-8
230 suppliers
$5.00/5mg
![Cyclohexanol, 1-[2-(dimethylamino)-1-[4-[(4-methoxyphenyl)methoxy]phenyl]ethyl]-](/CAS/20210305/GIF/1000377-24-1.gif)
1000377-24-1
0 suppliers
inquiry

93413-62-8
230 suppliers
$5.00/5mg

99300-78-4
601 suppliers
$5.00/25mg

93413-62-8
230 suppliers
$5.00/5mg
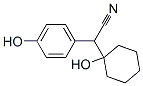
918344-20-4
21 suppliers
inquiry
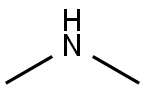
124-40-3
498 suppliers
$18.00/100ml

93413-62-8
230 suppliers
$5.00/5mg