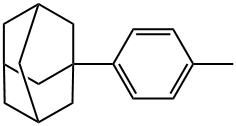
P-(1-ADAMANTYL)TOLUENE synthesis
- Product Name:P-(1-ADAMANTYL)TOLUENE
- CAS Number:1459-55-8
- Molecular formula:C17H22
- Molecular Weight:226.36
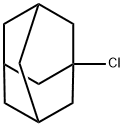
768-90-1
397 suppliers
$10.00/5g
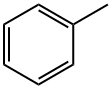
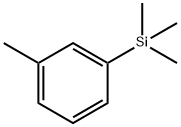
108-88-3
7 suppliers
$21.80/5 mL

1459-55-8
66 suppliers
$130.00/500mg
Yield:1459-55-8 99%
Reaction Conditions:
with molybdenum hexacarbonyl at 100; for 2 h;Catalytic behavior;Sealed tube;regioselective reaction;Reagent/catalyst;Temperature;Time;
Steps:
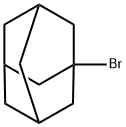
Alkylation of aromatic compound with 1-bromoadamantane. General procedure.
General procedure: Into a pressure microreactor of stainless steel of capacity 17 mL or into a glass ampule of capacity 10 mL (the results of parallel runs differed insignificantly) was charged 1-3 mmol of catalyst [Mo(CO)6, MoO2(acac)2, Cr(CO)6,CoBr2, CuBr, CuBr2, W(CO)6], 100 mmol of 1-bromoadamantane, and 500 mmol of aromatic compound, the reactor was hermetically closed (the ampule was sealed) and heated at 110-130° for 1-3 h. On completing the reaction the reactor (ampule) was cooled to room temperature, opened, the reaction mixture was neutralized with NaHCO3 and filtered through a bed of Al2O3. The unreacted aromatic compound was distilled off, the residue was distilled in propanol).
References:
Khusnutdinov;Shchadneva;Khisamova [Russian Journal of Organic Chemistry,2015,vol. 51,# 11,p. 1545 - 1550][Zh. Org. Khim.,2015,vol. 51,# 11,p. 1576 - 1581,6]

768-90-1
397 suppliers
$10.00/5g
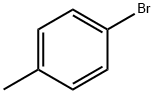
106-38-7
393 suppliers
$14.00/5g

1459-55-8
66 suppliers
$130.00/500mg
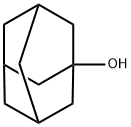
768-95-6
421 suppliers
$10.00/5g

108-88-3
7 suppliers
$21.80/5 mL

1459-55-8
66 suppliers
$130.00/500mg