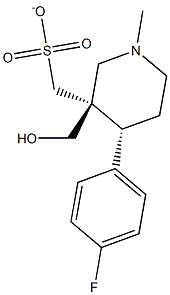
Paroxol Methanesulfonate synthesis
- Product Name:Paroxol Methanesulfonate
- CAS Number:608521-21-7
- Molecular formula:C14H20FNO3S
- Molecular Weight:301.38
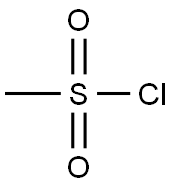
124-63-0
0 suppliers
$10.00/5g
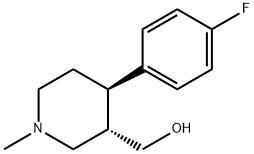
105812-81-5
306 suppliers
$9.00/1g

608521-21-7
15 suppliers
$180.00/50mg
Yield:608521-21-7 94%
Reaction Conditions:
with triethylamine in dichloromethane at 0 - 20; for 2 h;
Steps:
3
To a solution of trans-(4R,3S)-[4-(4-fluoro-phenyl)-1-methyl-piperidin-3-yl]-methanol (1.00 g, 4.48 mmol, 3B Medical Systems) in dry CH2Cl2 (7 mL) was added Et3N (1.5 mL). The mixture was cooled to 0° C. and treated with methanesulfonyl chloride (0.57 mL) and stirred in an ice bath for 0.5 hour and then for an additional 1.5 hours at ambient temperature. The reaction mixture was filtered through a cotton plug and washed through with additional dry CH2Cl2 (10 mL). The organic layer was diluted in a separatory funnel with ether (100 mL) and additional CH2Cl2 (20 mL) and washed three times with water (10 mL each time), dried (MgSO4), and concentrated to afford the desired product, methanesulfonic acid trans-(4R,3S)-4-(4-fluorophenyl)-1-methyl-piperidin-3-ylmethyl ester. Yield: 1.26 g (94%, 4.19 mmol). 1H-NMR (CDCl3) δ ppm: 1.79-1.90 (m, 2H); 1.91-2.37 (m, 4H); 2.38 (s, 3H); 2.87 (s, 3H); 2.95 (m, 1H); 3.12 (m, 1H); 3.81 (m, 1H); 3.93 (m, 1H); 7.01 (m, 2H); 7.16 (m, 2H).
References:
US2007/112031,2007,A1 Location in patent:Page/Page column 33
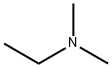
598-56-1
166 suppliers
$29.00/100mL
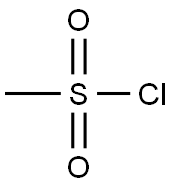
124-63-0
0 suppliers
$10.00/5g

105812-81-5
306 suppliers
$9.00/1g

608521-21-7
15 suppliers
$180.00/50mg