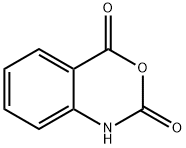
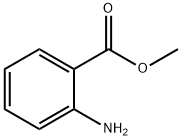
pentyl anthranilate synthesis
- Product Name:pentyl anthranilate
- CAS Number:30100-15-3
- Molecular formula:C12H17NO2
- Molecular Weight:207.27
Yield:30100-15-3 60%
Reaction Conditions:
with sodium hydroxide at 85;
Steps:
4 2.2.4 Pentyl-2-aminobenzoate (5)
Isatoic anhydride (8.16g, 50mmol) was dissolved in 40mL 1-pentanol and 0.1g NaOH (2.5mmol) was added. The mixture was heated at 85°C until the gas generation had ceased, subsequently cooled and poured in 120mL water. The product settled out as immiscible oil on the surface. The oil was separated and distilled under vacuum over a 20cm Vigreux column. The pure product, 6.26g of a colorless oil (yield 60%) passed over at 147.5°C (5Torr). H NMR (500MHz, CDCl3) δ 0.93 (t, 3H, 3J=7.0Hz, 5’-H); 1.40 (m, 4 H, 3’-H, 4’-H); 1.76 (quintet, 2H, 2’-H); 4.26 (t, 2H, 3J=6.7Hz, 1’-H); 5.72 (br s, 2H, NH); 6.64 (m, 2H, 3-H, 5-H); 7.25 (m, 1H, 4-H); 7.87 (d, 1H, 3J=8.3Hz, 6-H); C NMR (125MHz, CDCl3) δ 14.1 (C5’); 22.5 (C4’); 28.3 (C3’); 28.6 (C2’); 64.6 (C1’); 111.2 (C1); 116.3 (C3); 116.7 (C5); 131.3 (C6); 134.0 (C4); 150.6 (C2); 168.3 (C7); EI-MS (70eV): m/z (rel. int.)=207 [M+] (100), 193 (6), 137 (66), 119 (75), 92 (21); HR-EI-MS: m/z=207.1257; C12H17NO2 [M+] requires 207.1259.
References:
Roell, Daniela;R?sler, Thomas W.;Hessenkemper, Wiebke;Kraft, Florian;Hauschild, Monique;Bartsch, Sophie;Abraham, Tsion E.;Houtsmuller, Adriaan B.;Matusch, Rudolf;van Royen, Martin E.;Baniahmad, Aria [Journal of Steroid Biochemistry and Molecular Biology,2019,vol. 188,p. 59 - 70]