
PF-04457845 synthesis
- Product Name:PF-04457845
- CAS Number:1020315-31-4
- Molecular formula:C23H20F3N5O2
- Molecular Weight:455.43
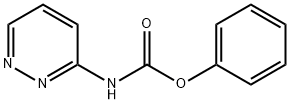
1020327-61-0
3 suppliers
inquiry
![Pyridine, 2-[3-(4-piperidinylidenemethyl)phenoxy]-5-(trifluoromethyl)-, hydrochloride (1:1)](/CAS/20210111/GIF/1020325-53-4.gif)
1020325-53-4
2 suppliers
inquiry

1020315-31-4
63 suppliers
$45.00/1mg
Yield: 86%
Reaction Conditions:
with N-ethyl-N,N-diisopropylamine in acetonitrile at 20; for 19 h;Product distribution / selectivity;
Steps:
5.b
Example 5b Large scale synthesis of N-pyridazin-3-yl-4-(3-{[5-(trifluoromethyl)pyridin-2-yl]oxy}benzylidene)piperidine-1-carboxamide To a mixture of 2-(3-piperidin-4-ylidenemethyl-phenoxy)-5-trifluoromethyl-pyridine, hydrochloride (37.1 g, 0.10 mol, see Example 1b, step 5) and phenyl pyridazin-3-ylcarbamate (21.5 g, 0.10 mol, see Example 39, steps 1 and 2) in acetonitrile (400 mL) was added dropwise diisopropylethyl amine (25.8 g, 0.20 mol). A solution formed after 2 h of stirring. The slightly turbid solution was stirred at ambient temperature for 17 h. It was poured into 2.5 L of stirred ice-water. The resulting mixture was stirred for 1 h. The solid was filtered off, rinsed with 300 mL of water and pressed dry under suction. This was dissolved in 400 mL of dichloromethane. Water was removed using a sep funnel and then the solution was dried over magnesium sulfate and concentrated in vacuo to ~50 mL. The viscous solution was diluted with 65 mL of ethyl acetate and then with 85 mL of methyl t-butyl ether. A solution formed, and then a solid began separating. The crystallizing mixture was kept at -10° C. for 2 h and filtered. The solid was rinsed with EtOAc:MTBE (40 mL) and pressed dry under suction. Further drying in vacuo at 40° C. for 7 h provided 30.3 g (66%) of product. The mother liquor was concentrated in vacuo to 19 g of a viscous oil. This was dissolved in 15 mL of ethyl acetate. The solution was diluted with 60 mL of methyl t-butyl ether, seeded and kept at 5° C. for 18 h. The solid which crystallized was filtered off, rinsed with 10 mL of methyl t-butyl ether, and pressed dry under suction. 9.0 g (20%) of additional product was obtained. Total yield=39.3 g (86%).
References:
FAY, Lorraine Kathleen;Johnson, Douglas S.;Kesten, Suzanne Ross;Lazerwith, Scott E.;Morris, Mark Anthony;Stiff, Cory Michael;Meyers, Marvin Jay;Wang, Lijuan Jane US2008/261941, 2008, A1 Location in patent:Page/Page column 19
![Pyridine, 2-[3-(4-piperidinylidenemethyl)phenoxy]-5-(trifluoromethyl)-, hydrochloride (1:1)](/CAS/20210111/GIF/1020325-53-4.gif)
1020325-53-4
2 suppliers
inquiry
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

1020315-31-4
63 suppliers
$45.00/1mg
![Pyridine, 2-[3-(chloroMethyl)phenoxy]-5-(trifluoroMethyl)-](/CAS/20150408/GIF/1020325-30-7.gif)
1020325-30-7
10 suppliers
inquiry

1020315-31-4
63 suppliers
$45.00/1mg
![BenzeneMethanol, 3-[[5-(trifluoroMethyl)-2-pyridinyl]oxy]-](/CAS/20150408/GIF/1020325-22-7.gif)
1020325-22-7
6 suppliers
inquiry

1020315-31-4
63 suppliers
$45.00/1mg
![Phosphonic acid, P-[[3-[[5-(trifluoroMethyl)-2-pyridinyl]oxy]phenyl]Methyl]-, diethyl ester](/CAS/20150408/GIF/1020325-38-5.gif)
1020325-38-5
2 suppliers
inquiry

1020315-31-4
63 suppliers
$45.00/1mg