
PHENYLGLYOXAL synthesis
- Product Name:PHENYLGLYOXAL
- CAS Number:1074-12-0
- Molecular formula:C8H6O2
- Molecular Weight:134.13
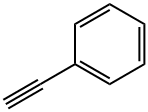
536-74-3
295 suppliers
$10.00/1g

1074-12-0
109 suppliers
$48.00/1g
Yield:1074-12-0 100%
Reaction Conditions:
with ruthenium trichloride;[bis(acetoxy)iodo]benzene in dichloromethane;water at 30; for 1 h;
Steps:
Typical method for isolation of aldehydes and diketone
General procedure: The typical solution containing 1 mmol of substrates, 0.1 mol % RuCl3 and 3 equiv of PhI(OAc)2, was heated and kept at constant 30°C. After Reaction for a period of time, the mixture was then separated with separatory funnel and the dichloromethane phase was dried over anhydrous Na2SO4, filtered and the solvent removed under vacuum. The crude product was purified via a standard silica gel chromatography using hexanes/ethyl acetate as eluent to give the desired aldehyde and diketone products. The isolated products were characterized by NMR (in CDCl3 with TMS as an internal standard) and ESI-MS. All the reactions were performed at least three times to establish there producibility and reliability.
References:
Mi, Chun;Li, Lu;Meng, Xiang-Guang;Yang, Ren-Qiang;Liao, Xiao-Hong [Tetrahedron,2016,vol. 72,# 42,p. 6705 - 6710] Location in patent:supporting information
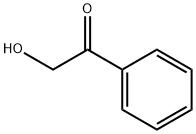
582-24-1
240 suppliers
$8.00/5g

1074-12-0
109 suppliers
$48.00/1g
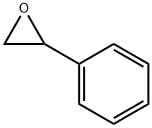
96-09-3
218 suppliers
$14.00/5g

1074-12-0
109 suppliers
$48.00/1g
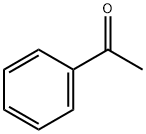
98-86-2
742 suppliers
$9.00/5g

1074-12-0
109 suppliers
$48.00/1g
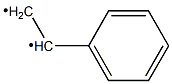
292638-84-7
0 suppliers
inquiry

1074-12-0
109 suppliers
$48.00/1g