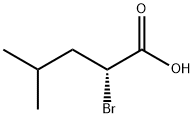
(R)-2-bromo-4-methylpentanoic acid synthesis
- Product Name:(R)-2-bromo-4-methylpentanoic acid
- CAS Number:42990-28-3
- Molecular formula:C6H11BrO2
- Molecular Weight:195.05
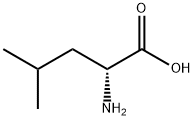
328-38-1
433 suppliers
$5.00/5g

42990-28-3
16 suppliers
inquiry
Yield:42990-28-3 99%
Reaction Conditions:
with sulfuric acid;sodium bromide;sodium nitrite in water at 0 - 20;
Steps:
General procedure for automated bromination of amino acid
General procedure: A mixture of amino acid (1.00 equiv.) and 30% sodium bromide aq. (1.30 mL/mmol) was added manually to RF1 and cooled to 0 oC. The solution was treated with 25% sulfuric acid aq. (1.30 mL/mmol, RR1) and 20% sodium nitrite aq. (0.650 mL/mmol, RR2) at 0 oC. After being stirred at the room temperature for 6 h, the aqueous layer was extracted with two portions of ethyl acetate. The combined organic layer was washed with brine, dried over MgSO4, filtered, and concentrated in vacuo. The residue was purified by column chromatography on silica gel to give α-bromocarboxylic acid. The spectral data of 2a1, 2b2, 2c3, 2d4, and 2e5 were identical to the reported data.
References:
Masui, Hisashi;Naito, Kohei;Minoshima, Mai;Kusayanagi, Akira;Yosugi, Sae;Shoji, Mitsuru;Takahashi, Takashi [Bioorganic and Medicinal Chemistry Letters,2021,vol. 40,art. no. 127961] Location in patent:supporting information

61-90-5
775 suppliers
$10.00/25G

42990-28-3
16 suppliers
inquiry
![1,2:5,6-Bis-O-(1-methylethylidene)-3-O-[4-methyl-1-[(trimethylsilyl)oxy]-1-penten-1-yl]-α-D-glucofuranose](/CAS/20210305/GIF/161661-14-9.gif)
161661-14-9
0 suppliers
inquiry

42990-28-3
16 suppliers
inquiry