
(S)-3-(3-METHOXYPHENYL)PIPERIDINE synthesis
- Product Name:(S)-3-(3-METHOXYPHENYL)PIPERIDINE
- CAS Number:88784-37-6
- Molecular formula:C12H17NO
- Molecular Weight:191.27
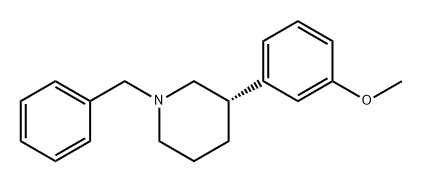
89874-44-2
0 suppliers
inquiry

88784-37-6
31 suppliers
$220.00/100mg
Yield:88784-37-6 94%
Reaction Conditions:
with hydrogen;ammonium formate;palladium on activated charcoal in methanol; for 1 h;Heating / reflux;
Steps:
S-3- (3-Methoxy-phenyl) -piperidine (S-9) l-Benzyl-3- (3- methoxy-phenyl) -piperidine (S-8) (306 mg, 1.09 mmol) was added to Pd/C (250 mg) , ammoniumformate (270 mg, 62.06 mmol) and 125 mL of methanol. The reaction mixture was brought to reflux while stirring in a nitrogen atmosphere for 1 h. Then the reaction mixture was left to cool to room temperature. The reaction mixture was filtered. To the filtrate some methanol was added and refluxed shortly. Again, this was filtered. Both methanol fractions were combined and the solvent was evaporated. Yield: 196 mg (94%). GCMS (EI): m/z 191 (M+), 162 (M-29); GC (100-3200C, 15°C/min): 4.3 min.
References:
WO2008/127188,2008,A1 Location in patent:Page/Page column 25

79601-21-1
34 suppliers
$155.00/50mg

88784-37-6
31 suppliers
$220.00/100mg

103291-08-3
5 suppliers
inquiry

79601-21-1
34 suppliers
$155.00/50mg

88784-37-6
31 suppliers
$220.00/100mg
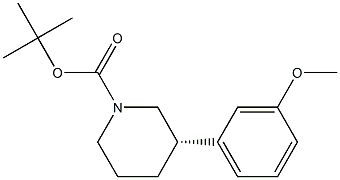
450416-70-3
0 suppliers
inquiry

88784-37-6
31 suppliers
$220.00/100mg

65292-99-1
36 suppliers
$47.00/100mg

88784-37-6
31 suppliers
$220.00/100mg