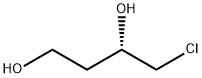
(S)-4-Chloro-1,3-butanediol synthesis
- Product Name:(S)-4-Chloro-1,3-butanediol
- CAS Number:139013-68-6
- Molecular formula:C4H9ClO2
- Molecular Weight:124.57
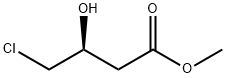
86728-93-0
78 suppliers
$55.00/250mg

139013-68-6
60 suppliers
inquiry
Yield:139013-68-6 98%
Reaction Conditions:
Stage #1:methyl (S)-4-chloro-3-hydroxybutanoate with sodium tetrahydroborate in methanol;toluene at 20; for 13 h;
Stage #2: with hydrogenchloride;water in methanol;toluene at 10;Product distribution / selectivity;
Steps:
16
Example 16; [90] 61.5 g (1.626 mol) of sodium borohydride was dissolved in 694 mL of toluene, and275.6 g (1.807 mol, 99.3% ee) of methyl (S)-4-chloro-3-hydroxybutyrate was added. After adding 52 g (1.626 mol) of methanol drop wise at room temperature for 1 hour, stirring was performed at room temperature for 12 hours. The reaction mixture was cooled below 1O0C and, after adding 183 g of 36% HCl dropwise, the solvent was removed by distillation under reduced pressure below 4O0C. Using 800 mL of methanol, concentration under reduced pressure was performed below 4O0C for 3 times. 800 mL of dichloromethane was added to the resultant residue. After filtering off solid inorganic materials, the solvent was removed under reduced pressure to obtain 220 g of (S)-4-chloro-l,3-butanediol as oil (yield = 98%).
References:
RSTECH CORPORATION WO2008/93955, 2008, A1 Location in patent:Paragraph 89-90
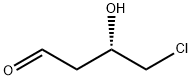
125781-04-6
9 suppliers
inquiry

139013-68-6
60 suppliers
inquiry