
tert-butyl 3,5-dioxopiperazine-1-carboxylate synthesis
- Product Name:tert-butyl 3,5-dioxopiperazine-1-carboxylate
- CAS Number:501127-89-5
- Molecular formula:C9H14N2O4
- Molecular Weight:214.22

24424-99-5
824 suppliers
$13.50/25G
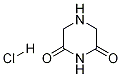
35975-30-5
22 suppliers
inquiry

501127-89-5
36 suppliers
$310.00/500mg
Yield:501127-89-5 78%
Reaction Conditions:
Stage #1: di-tert-butyl dicarbonate;piperazine-2,6-dione hydrochloridewith triethylamine in tetrahydrofuran;methanol;N,N-dimethyl-formamide; for 17 h;
Stage #2: with hydrogenchloride in tetrahydrofuran;methanol;N,N-dimethyl-formamide;
Steps:
39.3 3)
3) 4-tert-Butoxycarbonylpiperazine-2,6-dione The above piperazine-2,6-dione hydrochloride (1.58 g) was suspended in tetrahydrofuran (15 mL), methanol (15 mL), and N,N-dimethylformamide (30 mL), and, at room temperature, triethylamine (3.21 mL) and di-tert-butyl dicarbonate (2.51 g) were added thereto, followed by stirring for 17 hours. The reaction solvent was evaporated under reduced pressure, and water, 1M aqueous hydrochloric acid, and ethyl acetate were added to the residue for partitioning the residue. The aqueous layer was extracted with ethyl acetate, and the organic layers were combined, followed by washing with saturated brine and drying over sodium sulfate anhydrate. After a filtration step, the solvent was evaporated under reduced pressure, to thereby give 4-tert-butoxycarbonylpiperazine-2,6-dione as a solid product (1.75 g, 78%). 1H-NMR(400MHz,CDCl3)δ:1.48(9H,s), 4.30(2H,s), 8.14(1H,br).
References:
EP1762568,2007,A1 Location in patent:Page/Page column 56
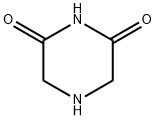
4774-22-5
85 suppliers
$20.00/250mg

24424-99-5
824 suppliers
$13.50/25G

501127-89-5
36 suppliers
$310.00/500mg