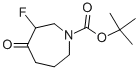
TERT-BUTYL 3-FLUORO-4-OXOAZEPANE-1-CARBOXYLATE synthesis
- Product Name:TERT-BUTYL 3-FLUORO-4-OXOAZEPANE-1-CARBOXYLATE
- CAS Number:644982-12-7
- Molecular formula:C11H18FNO3
- Molecular Weight:0
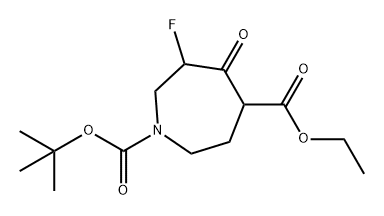
644982-11-6
1 suppliers
inquiry

644982-12-7
17 suppliers
$175.00/50mg
Yield:644982-12-7 21%
Reaction Conditions:
with water;lithium chloride in dimethyl sulfoxide at 125;
Steps:
51.B
The product from Step A was dissolved in DMSO (200 mL) and H2O (8.29 g, 460 mmol) followed by addition of LiCl (19.5 g, 460 mmol). The solution was warmed to 125 0C and the mixture was stirred overnight. The mixture was cooled to ambient temperature and partitioned between EtOAc (2.5 L) and water:brine (1 :1, 2.0 L). The mixture was filtered through activated charcoal over Celite, which was washed with EtOAc, forming visible layers. The organic layer was dried over Na2SO4, filtered and concentrated. The crude product was purified by column chromatography (10 to 80% EtOAc/hexanes) providing 4.5 g (21%) of the product as a thick pale orange oil.
References:
WO2010/22076,2010,A1 Location in patent:Page/Page column 79
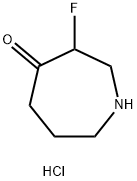
644982-13-8
13 suppliers
$350.00/100mg

24424-99-5
823 suppliers
$13.50/25G

644982-12-7
17 suppliers
$175.00/50mg