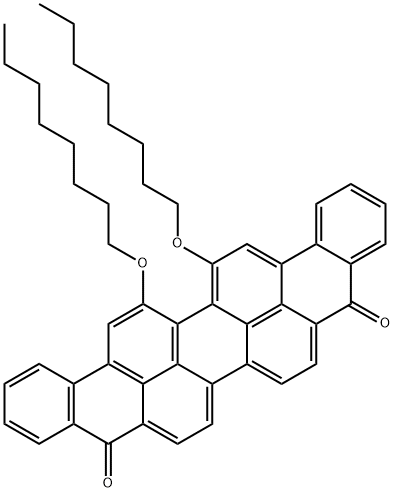
VIOLANTHRONE-79 synthesis
- Product Name:VIOLANTHRONE-79
- CAS Number:85652-50-2
- Molecular formula:C50H48O4
- Molecular Weight:712.91

111-83-1
491 suppliers
$12.00/25g
128-59-6
115 suppliers
$45.00/100mg

85652-50-2
87 suppliers
$12.00/250mg
Yield: 72%
Reaction Conditions:
with 18-crown-6 ether;potassium carbonate in 1-methyl-pyrrolidin-2-one at 100; for 8 h;Williamson Ether Synthesis;
Steps:
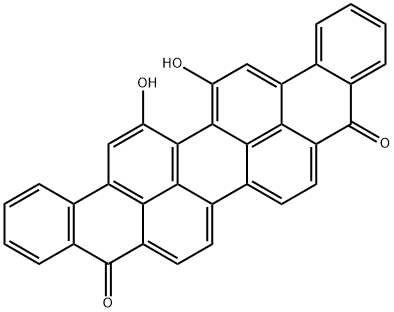
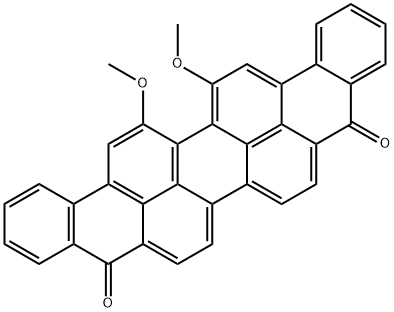
3 Implementing example three , 16, 17- two Xin Yangji Anthracyclinone purple preparation
Taking 1.21g (2.48mmol) 16, 17-dihydroxy Anthracyclinone purple puts in 100 ml flask, add 50mLN-methyl pyrrolidone, heating 10 min to 100 °C the 16, 17-dihydroxy Anthracyclinone purple disperse as much as possible. Then add 1.00g (7.2mmol) no water K2CO3, 0.98g (5.08mmol) 1-polybromide octane, and presence of 18-crown -6. Maintain reaction temperature 100 °C, reaction 8 hours the reaction is stopped.The reaction solution is poured into 100 ml water, fully mixing, filtering, drying to obtain the crude product. Dichloromethane is used for dissolving the crude product, filtering to remove undissolved components. turns on lathe does dichloromethane, to obtain the solid, dried to obtain 1.26g grams of product, yield is 72%. The silica gel chromatographic column, to CH2Cl2: CH3OH=20 1 as eluant, further elution separation and purification.
References:
Beijing University of Chemical Technology;Zhuang, Junpeng;Liu, Jie;Hao, Haijun;Niu, Junyu CN103804163, 2016, B Location in patent:Paragraph 0036; 0037; 0038
128-58-5
79 suppliers
$155.00/1g

85652-50-2
87 suppliers
$12.00/250mg