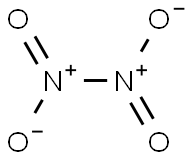
질소 테트라산화물
|
|
질소 테트라산화물 속성
- 녹는점
- −11 °C(lit.)
- 끓는 점
- 21 °C(lit.)
- 밀도
- 2.62 g/mL at 25 °C(lit.)
- 증기 밀도
- 1.58 (21 °C, vs air)
- 증기압
- 14.33 psi ( 20 °C)
- 용해도
- reacts with H2O
- 물리적 상태
- 가스
- 색상
- colorless liquid; equil with NO
- 수용성
- H2O [CRC10] 와 반응
- Dielectric constant
- 2.5(15℃)
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | T+ | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 26-34 | ||
| 안전지침서 | 9-26-28-36/37/39-45 | ||
| 유엔번호(UN No.) | UN 1067 2.3 | ||
| WGK 독일 | 1 | ||
| RTECS 번호 | QX1575000 | ||
| HS 번호 | 28112900 | ||
| 유해 물질 데이터 | 10544-72-6(Hazardous Substances Data) | ||
| 독성 | dog,LC50,inhalation,260mg/m3 (260mg/m3),CARDIAC: OTHER CHANGESLUNGS, THORAX, OR RESPIRATION: ACUTE PULMONARY EDEMABEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD,"Spravochnik po Toksikologii i Gigienicheskim Normativam Vol. -, Pg. 6, 1999. | ||
| 기존화학 물질 | KE-11944 |
질소 테트라산화물 C화학적 특성, 용도, 생산
개요
Nitrogen tetraoxide is primarily known for its use as a rocket propellant and became an oxidizer of choice by the late 1950s. Nitrogen tetraoxide has been used as a propellant for missiles as well as for spacecraft beginning with its use in Titan rockets in the late 1950s.화학적 성질
col liquid [CRC10]물리적 성질
Colorless liquid or gas; exists in equilibrium with NO2; density 1.45 g/mL at 20°C; boils at 21.25°C; freezes at -9.35°C to a colorless diamagnetic solid; critical temperature 157.85°C; critical pressure 99.64 atm; critical volume 167 cm3/mol; reacts with water.용도
Nitrogen tetroxide is a solvent and a powerful and selective oxidizing agent. It’s adducts with organic solvents are used to synthesize nitrates of noble metals.정의
A colorless gas that becomes a pale yellow liquid below 21°C and solidifies below –11°C. On heating, the gas dissociates to nitrogen dioxide molecules:N2O4(g) = 2NO2(g)
This dissociation is complete at 140°C. Liquid dinitrogen tetroxide has good solvent properties and is used as a nitrating agent.
제조 방법
Nitrogen tetroxide always is formed along with nitrogen dioxide during preparation of the dioxide (See Nitrogen Dioxide.) Mixed oxides are produced by oxidation of nitric oxide (NO) in air, heating metal nitrates, or by metals reacting with nitric acids or nitrates.일반 설명
Red-brown liquid with a sharp, unpleasant chemical odor. Low-boiling (boiling point 21.15°C) and held as a liquid by compression. Density 1.448 g / cm3. Consists of an equilibrium mixture of brown NO2 (nitrogen dioxide) and colorless N2O4 (diNITROGEN DIOXIDE). Evolves poisonous brown vapors. Cylinders and ton containers may not be equipped with a safety relief device. Prolonged exposure of the containers to fire or heat may result in their violent rupturing and rocketing.공기와 물의 반응
Reacts with water to form nitric acid and nitric oxide.위험도
See Nitrogen Dioxide, Hazard.건강위험
Very concentrated fumes produce coughing, choking, headache, nausea, pain in chest and abdomen; otherwise, few symtoms appear at time of exposure. After symptom-free period of 5-72 hours, pulmonary edema gradually develops, causing fatigue, restlessness, coughing, difficulty in breathing, frothy expectoration, mental confusion, lethargy, bluish skin, and weak, rapid pulse. Since NOX interferes with gas exchange in lungs, unconscious- ness and death by asphyxiation can result, usually within a few hours after onset of pulmonary edema.Safety Profile
A poison. Moderately toxic by inhalation. When heated to decomposition it emits toxic fumes of NOx. See also NITROGEN MONOXIDE.환경귀착
Nitrogen tetroxide is released into the atmosphere where it can undergo reactions, leading to air pollution.Purification Methods
Purify it by oxidation at 0o in a stream of oxygen until the blue colour changes to red-brown. Alternatively distil it from P2O5, then solidify it by cooling in a deep-freeze (at –78o, giving nearly colourless crystals). Oxygen can be removed by alternate freezing and melting. TOXIC VAPOUR. [Schenk in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I pp 488-489 1963.]질소 테트라산화물 준비 용품 및 원자재
원자재
준비 용품
질소 테트라산화물 관련 검색:
히드라진 수화물 산화 황 이산화질소 질소 테트라산화물
NITROUS OXIDE
NITRIC OXIDE
Nitrogen pentoxide
dinitrogen trioxide
nitrogen dioxide and nitric oxide mixture
Nitric oxide and dinitrogen tetroxide mixtures
Nitrogen Dioxide-15N,18O2
DETECTING TUBE FOR DETERMINATION OF NITROGEN DIOXIDE 5-50 MG/M3
PK10 NITROGEN DIOXIDE-0.5 DETECTOR TUBES
NITROGEN-15N DIOXIDE