|
|
| | 1H-1,2,4-Triazole-1-carboximidamide hydrochloride Basic information |
| Product Name: | 1H-1,2,4-Triazole-1-carboximidamide hydrochloride | | Synonyms: | 1H-1,2,4-TRIAZOLE-1-CARBOXAMIDINE MONOHYDROCHLORIDE;1-CARBAMIDINO-1,2,4-TRIAZOLE;1H-1,2,4-Triazole-1-carboxamidine monohydrochloride ,98.5%;1-AMIDINO-1,2,4-TRIAZOLE HYDROCHLORIDE;TIMTEC-BB SBB004089;1h-1,2,4-triazole-1-carboxamidinehydrochloride;1H-1,2,4-TRIAZOLE-1-CARBOXAMIDINE MONOHY;(1H)-1,2,4-Triazole-1-carboxamidineHClpurum | | CAS: | 19503-26-5 | | MF: | C3H6ClN5 | | MW: | 147.57 | | EINECS: | 606-331-2 | | Product Categories: | | | Mol File: | 19503-26-5.mol |  |
| | 1H-1,2,4-Triazole-1-carboximidamide hydrochloride Chemical Properties |
| Melting point | 215-220 °C | | storage temp. | Inert atmosphere,Room Temperature | | Water Solubility | Soluble in water | | form | powder to crystal | | color | White to Almost white | | BRN | 9500271 | | InChI | InChI=1S/C3H5N5.ClH/c4-3(5)8-2-6-1-7-8;/h1-2H,(H3,4,5);1H | | InChIKey | JDDXNENZFOOLTP-UHFFFAOYSA-N | | SMILES | C(N1N=CN=C1)(N)=N.Cl | | CAS DataBase Reference | 19503-26-5 |
| Hazard Codes | Xn | | Risk Statements | 22-36-52/53-43 | | Safety Statements | 26-36/37-61 | | RIDADR | 2811 | | WGK Germany | 3 | | HazardClass | 6.1 | | PackingGroup | III | | HS Code | 29339900 |
| | 1H-1,2,4-Triazole-1-carboximidamide hydrochloride Usage And Synthesis |
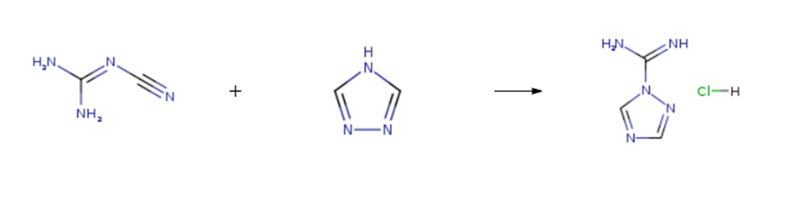
| Chemical Properties | White crystalline | | Uses | [1,2,4]Triazole-1-carboxamidine is an intermediate used in the synthesis of peramivir (P285500), a neuraminidase inhibitor developed as an antiviral agent for the treatment of influenza. | | Synthesis | 1H-1,2,4-Triazole-1-carboxamidine hydrochloride is prepared by the reaction of dicyandiamide and 1,2,4-Triazole. The steps are as follows:
Dissolve the compound (19.7 g, 0.29 mol) in 20 ml of tetrahydrofuran, and the temperature was raised to 70℃. A solution of dicyandiamide (20 g, 0.24 mol) was added and a solution of 4 mol / L HCl in tetrahydrofuran 27 g, 0.26 mol) at 70℃ for 2 h, the temperature was removed and cooled to room temperature. The solid was removed by filtration and 26.0 g was removed by filtration. The solvent was dried in vacuo at 45℃ to give a colorless oil which was recrystallized from methanol: dichloromethane = 2: 1 Of the mixed solution 50ml recrystallization, cooling crystallization, suction filter to get 6.2g solid, drying a total of 30.1g white powder. Yield 90.3%, purity 99,8%.
 |
| | 1H-1,2,4-Triazole-1-carboximidamide hydrochloride Preparation Products And Raw materials |
|