- Iodine pentoxide
-

- $1.00 / 1KG
-
2020-01-01
- CAS:12029-98-0
- Min. Order: 1KG
- Purity: 95-99%
- Supply Ability: 1ton
|
| Product Name: | Iodine pentoxide | | Synonyms: | Iodine pentoxid;Iodine(Ⅴ) oxide;di-Iodine pentoxide for analysis granular 0.5-2.5 mm EMSURE;Iodopentoxide 99.99% trace metals basis;iodineoxide(i2o5);IODINE(V) OXIDE;IODINE PENTOXIDE;IODINE PENTAOXIDE | | CAS: | 12029-98-0 | | MF: | I2O5 | | MW: | 333.81 | | EINECS: | 234-740-2 | | Product Categories: | Inorganics;halogen oxide | | Mol File: | 12029-98-0.mol | 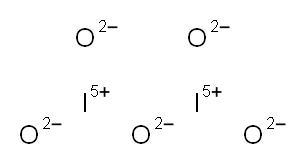 |
| | Iodine pentoxide Chemical Properties |
| Melting point | 300-350°C (dec.) | | density | 5.08 g/mL at 25 °C | | storage temp. | Store at +5°C to +30°C. | | solubility | insoluble in ethanol, ethyl ether, CS2 | | form | Crystalline | | Specific Gravity | 5.08 | | color | White to cream | | PH | 1.1 (100g/l, H2O, 25℃) | | Water Solubility | soluble | | Sensitive | Light Sensitive & Hygroscopic | | Merck | 14,5019 | | Stability: | Stability Strong oxidizer - contact with combustible material may cause fire. Light and moisture sensitive. Incompatible with organic materials, combustible materials, water. | | CAS DataBase Reference | 12029-98-0(CAS DataBase Reference) | | NIST Chemistry Reference | diiodine pentoxide(12029-98-0) | | EPA Substance Registry System | Iodine oxide (I2O5) (12029-98-0) |
| Hazard Codes | O,C | | Risk Statements | 8-34 | | Safety Statements | 17-26-27-36/37/39-45 | | RIDADR | UN 3085 5.1/PG 2 | | WGK Germany | 3 | | F | 8 | | TSCA | Yes | | HS Code | 2811 29 90 | | HazardClass | 5.1 | | PackingGroup | II |
| | Iodine pentoxide Usage And Synthesis |
| Physical Properties | White crystals; hygroscopic; density 4.98 g/cm3; decomposes around 300°C; highly soluble in water; soluble in nitric acid; insoluble in ethanol, ether and carbon disulfide.
| | Uses | Iodine pentoxide is used for analysis of carbon monoxide and for CO removal from air. It also is used as an oxidizing agent in other oxidation reactions.
| | Preparation | Iodine pentoxide is prepared by dehydration of iodic acid at 240°C.
2HIO3 → I2O5 + H2O
| | Reactions | Iodine pentoxide is a strong oxidizing agent and reacts with various oxidizable substances. It oxidizes carbon monoxide to carbon dioxide. The reaction is quantitative and used to measure carbon monoxide in the air:
I2O5 + 5CO → I2 + 5CO2
It reacts with hydrogen sulfide, forming sulfur dioxide:
3I2O5 + 5H2S → 3I2 + 5SO2 + 5H2O
Oxidation reaction also occurs with hydrogen chloride, metal hydrides and a number of metal salts. It dissolves in water reacting to form iodic acid:
I2O5 + H2O → 2HIO3
Dissociation into iodine and oxygen commences when heated above 270°C.
| | Chemical Properties | needle shaped crystals | | Physical properties | White crystals; hygroscopic; density 4.98 g/cm3; decomposes around 300°C;highly soluble in water; soluble in nitric acid; insoluble in ethanol, ether andcarbon disulfide. | | Uses | Iodine (V) oxide is used as an oxidizing agent to remove carbon monoxide from gas. | | Uses | Oxidizes carbon monoxide to carbon dioxide with the formn of iodine (Ditte's reaction, which proceeds rapidly at 65° or above, but slowly at room temp). This reaction is used in gas analysis and for removing carbon monoxide from the air (in respirators preferably in presence of some H2SO4). | | Definition | ChEBI: Diiodine pentaoxide is an iodine oxide. |
| | Iodine pentoxide Preparation Products And Raw materials |
|