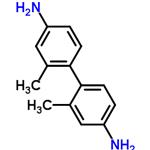
- m-tolidine
-
- $158.00 / 1KG
-
2024-01-06
- CAS:84-67-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: g-kg-tons, free sample is available
|
| | m-Tolidine Basic information |
| Product Name: | m-Tolidine | | Synonyms: | 2,2'-dimethyl[1,1'-biphenyl]-4,4'-diamine;4-(4-amino-2-methylphenyl)-3-methylbenzenamine;4,4'-Diamino-2,2'-dimethylbiphenyl;2,2'-DIMETHYL-4,4'-DIAMINOBIPHENYL;APB-144;M-TOLIDINE;1’-Biphenyl]-4,4’-diamine,2,2’-dimethyl-[1;2,2’-dimethylbenzidine | | CAS: | 84-67-3 | | MF: | C14H16N2 | | MW: | 212.29 | | EINECS: | 201-551-1 | | Product Categories: | Intermediates of Dyes and Pigments;fine chemicals, specialty chemicals, intermediates, electronic chemical, organic synthesis;fine chemicals, specialty chemicals, intermediates, electronic chemical, organic synthesis, functional materials;84-67-3 | | Mol File: | 84-67-3.mol |  |
| | m-Tolidine Chemical Properties |
| Melting point | 105-106℃ | | Boiling point | 344.4±37.0 °C(Predicted) | | density | 1.106 | | storage temp. | Keep in dark place,Sealed in dry,Room Temperature | | solubility | Chloroform (Slightly), Methanol (Slightly) | | form | Solid | | pka | 4.89±0.10(Predicted) | | color | White to Off-White | | InChI | InChI=1S/C14H16N2/c1-9-7-11(15)3-5-13(9)14-6-4-12(16)8-10(14)2/h3-8H,15-16H2,1-2H3 | | InChIKey | QYIMZXITLDTULQ-UHFFFAOYSA-N | | SMILES | C1(C2=CC=C(N)C=C2C)=CC=C(N)C=C1C | | CAS DataBase Reference | 84-67-3 |
| Safety Statements | 24/25 | | HS Code | 29215900 |
| | m-Tolidine Usage And Synthesis |
| Chemical Properties | m-Tolidine is pink particles and can form prisms when exposed to water. It is soluble in cold water, alcohol, and ether. In the presence of iron(III) chloride, m-Tolidine does not produce any color. It is tetrazotized with nitrite and acid and couples to form dyes. | | Uses | 2,2''-Dimethyl-[1,1''-biphenyl]-4,4''-diamine (cas# 84-67-3) is a useful reagent for manufacturing cured films and semiconductor devices. | | Uses | M-TOLIDINE is used for preparation polyimide and epoxy resin material. | | Preparation | M-Tolidine is made by the reduction of m-nitrotoluene with zinc dust and sodium hydroxide solution. The subsequent rearrangement is carried out with hydrochloric acid or sulfuric acid and gives rise to m-tolidine dihydrochloride or m-tolidine sulfate, which are isolated as salts. The free base is diffificult to isolate and is therefore not a commercial product. | | Definition | M-Tolidine gives no color in the presence of iron(III) chloride. It is tetrazotized with nitrite and acid and couples to form dyes. M-Tolidine was used in the past for the production of azo dyes. | | Chemical Reactivity | M-Tolidine gives no color in the presence of iron(III) chloride. It is tetrazotized with nitrite and acid and couples to form dyes. |
| | m-Tolidine Preparation Products And Raw materials |
| Raw materials | Phenylboronic acid pinacol ester-->Sodium hydroxide-->Hydrochloric acid-->Bis(pinacolato)diboron-->XPhos Pd G2-->2-IODO-5-NITROTOLUENE-->3-Nitrotoluene-->Zinkamalgam | | Preparation Products | 4,4'-Diiodo-2,2'-dimethylbiphenyl-->2-Naphthalenesulfonic acid, 5,5'-[(2,2'-dimethyl[1,1'-biphenyl]-4,4'-diyl)bis(azo)]bis[6-hydroxy-, disodium salt-->1,3-Naphthalenedisulfonic acid, 8-[[2,2'-dimethyl-4'-[[2-oxo-1-[(phenylamino)carbonyl]propyl]azo][1,1'-biphenyl]-4-yl]azo]-7-hydroxy-, disodium salt-->2,7-Naphthalenedisulfonic acid, 3-[[3,3'-dimethyl-4'-[[3-oxo-1-[(phenylamino)carbonyl]propyl]azo][1,1'-biphenyl]-4-yl]azo]-4-hydroxy-, disodium salt-->disodium 4-hydroxy-3-[[4'-[(2-hydroxynaphthyl)azo]-2,2'-dimethyl[1,1'-biphenyl]-4-yl]azo]naphthalene-2,7-disulphonate-->C.I. Acid Orange 49-->Acid Red 111-->sodium 4-chloro-3-[4-[[2,2'-dimethyl-4'-[[4-[(phenylsulphonyl)oxy]phenyl]azo][1,1'-biphenyl]-4-yl]azo]-4,5-dihydro-3-methyl-5-oxo-1H-pyrazol-1-yl]benzenesulphonate-->C.I. Acid black 70-->Acid Yellow 86 |
|