OXOMEMAZINE manufacturers
- Oxomemazine
-

- $200.00 / 1kg
-
2023-06-26
- CAS:3689-50-7
- Min. Order: 1kg
- Purity: 99%
- Supply Ability: 1000kg/Month
- Oxomemazine
-
- $100.00 / 1g
-
2023-04-11
- CAS:3689-50-7
- Min. Order: 10g
- Purity: 99%
- Supply Ability: 1000g
|
| | OXOMEMAZINE Basic information |
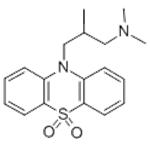
| Product Name: | OXOMEMAZINE | | Synonyms: | OXOMEMAZINE;10-(3-(Dimethylamino)-2-methylpropyl)phenothiazine-5,5-dioxide;10H-Phenothiazine-10-propanamine, N,N,beta-trimethyl-, 5,5-dioxide;3-(5,5-Dioxido-10H-phenothiazin-10-yl)-N,N,2-trimethyl-1-propanamine;6487 RP;6847 R.P.;6847 RP;Alimemazine S,S-dioxide | | CAS: | 3689-50-7 | | MF: | C18H22N2O2S | | MW: | 330.44 | | EINECS: | 222-996-8 | | Product Categories: | | | Mol File: | 3689-50-7.mol | 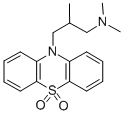 |
| | OXOMEMAZINE Chemical Properties |
| Melting point | 115° | | Boiling point | 492.4±34.0 °C(Predicted) | | density | 1.1453 (rough estimate) | | refractive index | 1.6000 (estimate) | | storage temp. | Store at -20°C | | solubility | ≥ 33mg/mL in DMSO | | pka | 9.31±0.28(Predicted) |
| Toxicity | LD50 oral in mouse: 140mg/kg |
| | OXOMEMAZINE Usage And Synthesis |
| Originator | Doxergan,Specia,France,1964 | | Uses | (±)-Oxomemazine, is a phenothiazine-type antihistamines, that has also shown antibacterial activity against Mycobacterium tuberculosis. | | Definition | ChEBI: Oxomemazine is a member of phenothiazines. | | Manufacturing Process | Phenothiazine is reacted with 3-dimethylamino-2-methylpropyl chloride in the presence of sodium amide to give 3-(10-phenthiazinyl)-2-methyl-1dimethylaminopropane. 11.9 g of of this intermediate is dissolved with agitation in glacial acetic acid (120 cc). Pure sulfuric acid (d = 1.83; 0.5 cc) is added and a mixture of glacial acetic acid (10 cc) and hydrogen peroxide (8.5 cc of a solution containing 38 g of hydrogen peroxide in 100 cc) is then run in over 20 minutes. The temperature rises from 25°C to 35°C and is then kept at 60°C for 18 hours. The mixture is cooled and water (150 cc) is added and, with cooling, aqueous sodium hydroxide (d = 1.33; 220 cc). The resulting
mixture is extracted with ethyl acetate (3 x 100 cc), the solvent is evaporated on a water bath and the residue is recrystallized from heptane (150 cc). 3(9,9-dioxy-10-phenthiazinyl)-2-methyl-1-dimethylaminopropane (78 g) is obtained, MP 115°C.
The corresponding hydrochloride prepared in ethyl acetate and recrystallized from a mixture of ethanol and isopropanol melts at 250°C. | | Therapeutic Function | Antihistaminic |
| | OXOMEMAZINE Preparation Products And Raw materials |
|