|
|
| | N-(PHENYLTHIO)PHTHALIMIDE Basic information |
| | N-(PHENYLTHIO)PHTHALIMIDE Chemical Properties |
| Melting point | 160-163 °C(lit.) | | Boiling point | 432.2±28.0 °C(Predicted) | | density | 1.42±0.1 g/cm3(Predicted) | | storage temp. | Sealed in dry,Room Temperature | | solubility | DMSO: soluble25mg/mL, clear, yellow | | pka | -3.05±0.20(Predicted) | | form | powder to crystal | | color | White to Light yellow | | CAS DataBase Reference | 14204-27-4 |
| Safety Statements | 24/25 | | WGK Germany | 3 | | HS Code | 29309090 |
| | N-(PHENYLTHIO)PHTHALIMIDE Usage And Synthesis |
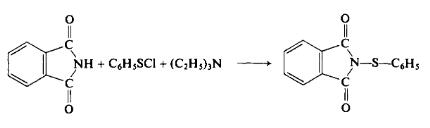
| Preparation | (a) Preparation of benzenesulfenyl chloride. To a stirred solution of 55 gm (0.5 mole) of benzene thiol in 300 ml of η -pentane at 0°C is added chlorine gas until an assay (GC) of the resulting red-orange solution shows quantitative conversion to the sulfenyl chloride. The reaction usually requires about 39 gm (0.6 mole) of chlorine.
(b) Reaction of benzenesulfenyl chloride with phthalimide. To a stirred solution of 73.5 gm (0.5 mole) of phthalimide in 200 ml of dimethylforma-mide is first added 60 gm (0.6 mole) of triethylamine. Then the sulfenyl chloride solution from (a) is slowly added dropwise. The reaction mixture is stirred for \ hr, poured into 2 liters of cold water, filtered, and dried to afford 121 gm (95%), m.p. 160-161°C (recrystallized from ethanol).
Complex imides such as camphorimide and 9,10-dihydroanthracene-9,10-endo-a,/3-succinimide have been alkylated in methylene dichloride with alkyl halides, using tetrabutylammonium bromide as a phase-transfer catalyst and aqueous potassium hydroxide as a base.
 |
| | N-(PHENYLTHIO)PHTHALIMIDE Preparation Products And Raw materials |
|