|
|
| | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE) Basic information |
| Product Name: | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE) | | Synonyms: | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE);TRIFLUOROMETHANESULFONIC ACID DIISOPROPYLSILYLENE ESTER;DIISOPROPYLSILYL BIS(TRIFLUOROMETHANE-SU LFONATE), TECH.;diisopropylbis(trifluoromethanesulfonyl)silane;Bis(trifluoromethanesulfonic acid)diisopropylsilanediyl ester;Diisopropylsilylenebis(trifluoromethanesulfonate);Methanesulfonic acid, trifluoro-, bis(1-methylethyl)silylene ester;Methanesulfonic acid, 1,1,1-trifluoro-, bis(1-methylethyl)[[(trifluoromethyl)sulfonyl]oxy]silyl ester | | CAS: | 85272-30-6 | | MF: | C8H14F6O6S2Si | | MW: | 412.4 | | EINECS: | | | Product Categories: | | | Mol File: | 85272-30-6.mol |  |
| | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE) Chemical Properties |
| Boiling point | 85-86 °C2 mm Hg(lit.) | | density | 1.396 g/mL at 25 °C(lit.) | | refractive index | n20/D 1.382(lit.) | | Fp | >230 °F | | Specific Gravity | 1.396 | | Hydrolytic Sensitivity | 8: reacts rapidly with moisture, water, protic solvents | | BRN | 3567500 |
| Hazard Codes | C | | Risk Statements | 34-37 | | Safety Statements | 26-36/37/39-45 | | RIDADR | UN 3265 8/PG 2 | | WGK Germany | 3 | | F | 10-21 | | HazardClass | 8 | | PackingGroup | III |
| | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE) Usage And Synthesis |
| Physical properties | bp 85–86°C/2mmHg;d 1.396 g cm?3; fp 110°C. | | Uses | Diisopropylsilyl
bis(trifluoromethanesulfonate) has more recently been
used to prepare diisopropylsilyl-linked oligonucleotide analogs.
Silylation of 5'-O-(dimethoxytrityl)-2'-deoxynucleosides using
diisopropylsilyl bis(trifluoromethanesulfonate) and the hindered
base 2,6-di-t-butyl-4-methylpyridine (Dtbp) provided quantitative
yield of 3'-O-diisopropylsilanols (eq 2). Hence, silylation
of 5'-O-(dimethoxytrityl)thymidine, N6-benzoyl-2'-deoxy-5'-O-
(dimethoxytrityl)adenosine, N4-benzoyl-2'-deoxy-5'-O-(dimethoxytrityl)
cytidine, and N2-isobutyryl-2'-deoxy-5'-O-(dimethoxytrityl)
guanosine gave the desired products 5a–d in excellent
yields.
Coupling of products 5a–d with unprotected thymidine
resulted in formation of 3',5'-linked dinucleosides 6a–d selectively,
and the dimer 7 from reaction with secondary hydroxyl
group of thymidine was not observed (eq 3). Subsequent silylation
and coupling with another molecule of thymidine gave 3�,5�-
linked trinucleoside 8 in 76% yield (eq 4). A tetrathymidylate oligomer was prepared by 3'-O-silylation of 8 followed by
coupling with thymidine. Although no self-condensation was
observed, only 30% of the product 9 was obtained after purification
by preparative HPLC.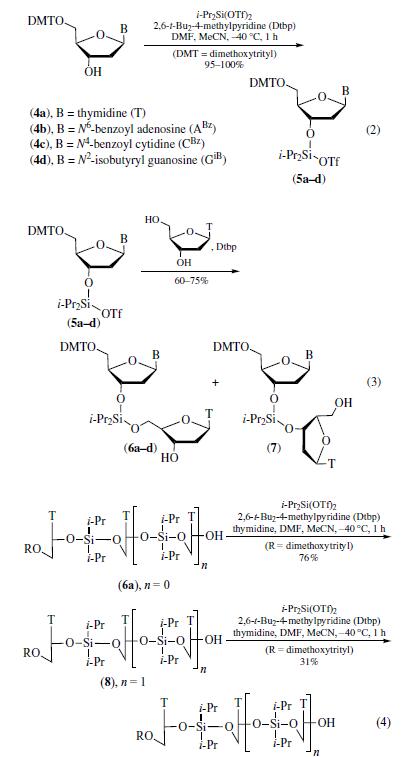 | | Preparation | conveniently prepared by the slow addition
of 2 equiv of trifluoromethanesulfonic acid to a stirring mixture
of chlorodiisopropylsilane, followed by direct distillation of the
product from the reaction flask to yield the product as a pale
yellow oil. |
| | DIISOPROPYLSILYL BIS(TRIFLUOROMETHANESULFONATE) Preparation Products And Raw materials |
|