|
|
| | 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester Basic information |
| Product Name: | 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester | | Synonyms: | 2-amino-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)nicotinonitrile;2-amino-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridine-3-carbonitrile;3-Pyridinecarbonitrile, 2-amino-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)- | | CAS: | 1246372-66-6 | | MF: | C12H16BN3O2 | | MW: | 245.09 | | EINECS: | | | Product Categories: | | | Mol File: | 1246372-66-6.mol |  |
| | 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester Chemical Properties |
| storage temp. | Keep in dark place,Inert atmosphere,Room temperature | | InChI | InChI=1S/C12H16BN3O2/c1-11(2)12(3,4)18-13(17-11)9-5-8(6-14)10(15)16-7-9/h5,7H,1-4H3,(H2,15,16) | | InChIKey | KRNXYPYYVHFVQS-UHFFFAOYSA-N | | SMILES | C1(N)=NC=C(B2OC(C)(C)C(C)(C)O2)C=C1C#N |
| | 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester Usage And Synthesis |
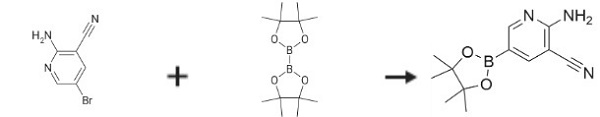
| Synthesis | 2-amino-5-bromonicotinonitrile (50 g, 0.224 mol), bis (pinacolato) diboron (85.6 g, 0.337 mol), KOAc (44.1 g, 0.449 mol) and Pd (dppf) Cl2. CH2C12 (2.77 g, 3.4 mmol) was charged into a flask. Dioxane (400 mL) was added. The reaction mixture was stirred at 100 ℃ for 2 h under Ar. When LC-MS indicated that the reaction was completed, the mixture was cooled to room temperature. Subsequently, the mixture was filtered through diatomite, concentrated, diluted with a mixture of ethyl acetate and hexane (3:1, 1000 mL), filtered through silica gel (300-400 mesh), concentrated, crystallized and dried to give 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester (32 g, 66%) as a white solid.
 |
| | 2-Amino-3-cyanopyridine-5-boronic Acid Pinacol Ester Preparation Products And Raw materials |
|