| Company Name: |
Hubei Jusheng Technology Co.,Ltd. |
| Tel: |
18871490254 |
| Email: |
linda@hubeijusheng.com |
| Products Intro: |
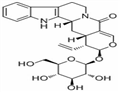
Product Name:(1R)-1β-Ethenyl-2α-(β-D-glucopyranosyloxy)-1,2,7,8,13,13bα,14,14aα-octahydro-5H-indolo[2,3-a]pyrano[3,4-g]quinolizine-5-one
CAS:23141-27-7
Purity:0.99 Package:5KG;1KG
|
|
|
|
|
| Company Name: |
BOC Sciences |
| Tel: |
+1-631-485-4226 |
| Email: |
inquiry@bocsci.com |
| Products Intro: |
Product Name:(1R)-1β-Ethenyl-2α-(β-D-glucopyranosyloxy)-1,2,7,8,13,13bα,14,14aα-octahydro-5H-indolo[2,3-a]pyrano[3,4-g]quinolizine-5-one
CAS:23141-27-7
Purity:98.5% Package:1mg Remarks:BOC Sciences also provides custom synthesis services for (1R)-1β-Ethenyl-2α-(β-D-glucopyranosyloxy)-1,2,7,8,13,13bα,14,14aα-octahydro-5H-indolo[2,3-a]pyrano[3,4-g]quinolizine-5-one
|
|
|
|
|
- Vincosamide
-
- $0.00 / 5mg
-
2023-02-24
- CAS:23141-27-7
- Min. Order: 5mg
- Purity: ≥98%(HPLC)
- Supply Ability: 10 g
|
| | Vincosamide Basic information |
| Product Name: | Vincosamide | | Synonyms: | (3β,15β)-19,20-Didehydro-16α-ethenyl-17β-(β-D-glucopyranosyloxy)-18-oxayohimban-21-one;Camptoside;Oxayohimban-21-one,19,20-didehydro-16-ethenyl-17-(b-D-glucopyranosyloxy)-, (3b,15b,16a,17b)-;Oxayohimban-21-one, 19,20-didehydro-16-ethenyl-17-(β-D-glucopyranosyloxy)-, (3β,15β,16α,17β)-;Vincosamide >=90% (LC/MS-ELSD);(3β,15β,16α,17β)-21-Oxo-16-vinyl-19,20-didehydro-18-oxayohimban-17-ylβ-D-glucopyranoside;Vincoside lactam;Vincosamide | | CAS: | 23141-27-7 | | MF: | C26H30N2O8 | | MW: | 498.52 | | EINECS: | | | Product Categories: | | | Mol File: | 23141-27-7.mol |  |
| | Vincosamide Chemical Properties |
| | Vincosamide Usage And Synthesis |
| Description | The alkaloids vincosamide and strictosamide are an important class of indole ring- containing alkaloids. They were extracted and differentiated from the root bark of Nauclea latifolia belonging to the family Rubiaceae. Both the compounds were tested for anti-HSV-2 activity for both acyclovir-sensitive and acyclovir-resistant HSV-2 strains and showed good anti-HSV-2 potential. | | Uses | Vincosamide is an alkaloid from Psychotria leiocarpa extract, inhibits the acetylcholinesterase (AChE) activity with anti-inflammatory activity. Vincosamide can effect relaxation of the supercoiled pBR322 plasmid DNA in the presence of Cu2+. | | Definition | ChEBI: Vincosamide is a monoterpenoid indole alkaloid. | | References | [1] HUIHUI TI. Progress of Plant Medicine Derived Extracts and Alkaloids on Modulating Viral Infections and Inflammation.[J]. Drug Design, Development and Therapy, 2021: 1385-1408. DOI:10.2147/DDDT.S299120.
[2] ANELISE SAMARA NAZARI FORMAGIO. Psychotria leiocarpa Extract and Vincosamide Reduce Chemically-Induced Inflammation in Mice and Inhibit the Acetylcholinesterase Activity.[J]. Inflammation, 2019, 42 5: 1561-1574. DOI:10.1007/s10753-019-01018-w.
[3] MING YUE ZHU. Vincosamide Has a Function for Inhibiting Malignant Behaviors of Hepatocellular Carcinoma Cells.[J]. World Journal of Oncology, 2022, 13 5: 272-288. DOI:10.14740/wjon1514.
[4] SEGUN A. ADERIBIGBE . Bioactivity and cytotoxicity profiling of vincosamide and strictosamide, anthelmintic epimers from Sarcocephalus latifolius (Smith) Bruce leaf[J]. Journal of ethnopharmacology, 2021, 265: Article 113142. DOI:10.1016/j.jep.2020.113142.
[5] ZHU M, FENG H, LIN B, et al. Vincosamide inhibits malignant behaviors of hepatocellular carcinoma cells by activating caspase-3 activity and blocking the PI3K/AKT signaling pathway[C]. 2020. DOI:10.21203/rs.3.rs-28543/v1. |
| | Vincosamide Preparation Products And Raw materials |
|