|
|
| | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone Basic information |
| Product Name: | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone | | Synonyms: | (3R,4S)-1-Benzoyl-3-(1-Ethoxyethoxy)-4-Phenyl-2-AzetidinoneCasNo.;(3R,4S)-1-BENZOYL-3-(2-ETHOXYETHOXYL)-4-PHENYL-2-AZETIDINONE;(3R,4S)-3-(1-ethoxyethoxy)-4-phenyl-N-benzoyl-2-azetidinone;(3R,4S)-1-Benzoyl-3-(2-Ethoxye;DOCETAXEL SIDE CHAIN / (3R,4S)-1-BENZOYL-3-(2-ETHOXYETHOXY)-4-PHENYL-2-PHENYL-AZETIDIONE;(3R,4S)-1-BENZOYL-3-(2-ETHOXYETHOXYL) -4-PHENYL-2-AZETIDINONE 97%;SIDE CHAINS OF PACLITAXEL--1;(3R,4S)-1-benzoyl-3-(1-ethoxyethoxy)-4-phenylazetidin-2-one | | CAS: | 201856-53-3 | | MF: | C20H21NO4 | | MW: | 339.39 | | EINECS: | | | Product Categories: | Intermediates | | Mol File: | 201856-53-3.mol | 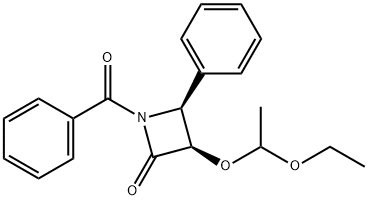 |
| | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone Chemical Properties |
| | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone Usage And Synthesis |
| Uses | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone is an investigational intermediate chemical for Paclitaxel and Docetaxel. | | Synthesis | To a solution of 2.35 g (10 mmol) of cis-3-(1-ethoxyethoxy)-4-phenylazetidin-2-one in 40 mL of THF at -78° C, was added 6.1 mL (10.07 mmol) of a 1.65M solution of n-butyllithium in hexane. The mixture was stirred for 10 min at -78° C and a solution of 1.42 g (10.1 mmol) of benzoyl chloride in 10 mL of THF was added. The mixture was stirred at -78° C for 1 h diluted with 70 mL of saturated aqueous sodium bicarbonate and extracted with three 50 mL portions of ethyl acetate. The combined ethyl acetate extracts were dried over sodium sulfate and concentrated to give 3.45 g of oil. Chromatography of the oil on silica gel eluted with ethyl acetate/hexane gave 3.22 g (95%) of (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone as a colorless oil. |
| | (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-azetidinone Preparation Products And Raw materials |
|