
cannabigerol synthesis
- Product Name:cannabigerol
- CAS Number:25654-31-3
- Molecular formula:C21H32O2
- Molecular Weight:316.48
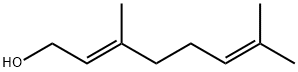
106-24-1
563 suppliers
$6.00/25g
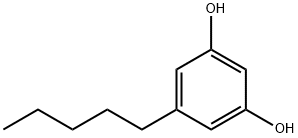
500-66-3
399 suppliers
$50.00/100mg

25654-31-3
69 suppliers
$47.00/1mg
Yield:25654-31-3 80%
Reaction Conditions:
with toluene-4-sulfonic acid in chloroform at 20; for 12 h;Darkness;
Steps:
2 Example 2: Synthesis of 2-[(2E)-3,7-dimethylocta-2,6-dienyl]-5-pentyl-benzene-1,3-diol (Cannabigerol (CBG)
Example 2: Synthesis of 2-[(2E)-3,7-dimethylocta-2,6-dienyl]-5-pentyl-benzene-1,3-diol (Cannabigerol (CBG) (0130) CBG was synthesized following the protocol disclosed by Taura et al., (1996), The Journal of Biological Chemistry, Vol. 271, No. 21, p. 17411-17416. (0131) Geraniol (3 g, 0.0194 mol) and olivetol (2 g, 0.0111 mol) were dissolved in 400 mL of chloroform containing 80 mg of p-toluenesulfonic acid as catalyst and the reaction mixture was stirred at room temperature for 12 h in the dark. After 12 hours, the reaction mixture was washed with saturated sodium bicarbonate (400 mL) and then with H2O (400 mL). The chloroform layer was concentrated at 40° C. under reduced pressure, and the residue obtained was chromatographed on a 2.0 cm×25 cm silica gel column using benzene (1000 mL) as the eluent to give 1.4 g (0.00442 mol) (39.9%) CBG as product. (0132) Alternatively crude CBG was purified as follows. To a 250 mL beaker was added 7.25 g crude CBG and 50 mL benzene. The flask was swirled to dissolve the CBG and 50 g silica gel was added, along with a stir bar. The solution was stirred overnight, and then poured into a 44 cm×2.75 cm column. The column was eluted with 300 mL benzene. The eluent, approximately 70 mL fractions were assayed for CBG. Fractions 1, 2, and 3 (230 mL) that contained CBG were combined and the solvent removed under pressure to give 6.464 g residue containing >80% CBG, having a purity suitable for use in the next synthetic step. (0133) In one embodiment, crude CBG was purified by mixing 7.25 g crude CBG residue with a slurry of silica gel (50 mL), in a 250 ml Beaker. This mixture was slowly agitated for 1 hour and then vacuum filtered using a fine mesh filter paper. The filter cake was washed with 250 ml benzene until a clear filtrate was obtained. The solvent from the filtrate was removed under reduced pressure to give 6.567 g of a residue having >80% CBG.
References:
US2017/283837,2017,A1 Location in patent:Paragraph 0130; 0131; 0132; 0133

6138-90-5
123 suppliers
$49.35/5g

500-66-3
399 suppliers
$50.00/100mg

25654-31-3
69 suppliers
$47.00/1mg
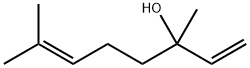
78-70-6
583 suppliers
$26.00/25mL

500-66-3
399 suppliers
$50.00/100mg

25654-31-3
69 suppliers
$47.00/1mg

106-24-1
563 suppliers
$6.00/25g

500-66-3
399 suppliers
$50.00/100mg
![1,3-Benzenediol, 4-[(2E)-3,7-dimethyl-2,6-octadien-1-yl]-5-pentyl-](/CAS/20210111/GIF/55854-24-5.gif)
55854-24-5
2 suppliers
inquiry

25654-31-3
69 suppliers
$47.00/1mg
![Benzoic acid, 3-[(2E)-3,7-dimethyl-2,6-octadien-1-yl]-2,4-dihydroxy-6-pentyl-, methyl ester](/CAS/20200611/GIF/63953-93-5.gif)
63953-93-5
0 suppliers
inquiry

25654-31-3
69 suppliers
$47.00/1mg