| Identification | More | [Name]
9-Bromoanthracene | [CAS]
1564-64-3 | [Synonyms]
9-BROMOANTHRACENE
9-BROMOANTHRENE
MS-BROMOANTHRACENE
RARECHEM AQ BD AN02
TIMTEC-BB SBB007978
9-Anthracenyl bromide
Anthracene, 9-bromo-
9-Bromoanthracene98%
9-Bromoanthracene 98%
9-BROMOANTHRACENE: TECH., 90%
10-Bromoanthracene
9-Anthryl bromide
9-Bromo anthrecene | [EINECS(EC#)]
216-359-3 | [Molecular Formula]
C14H9Br | [MDL Number]
MFCD00001243 | [Molecular Weight]
257.13 | [MOL File]
1564-64-3.mol |
| Chemical Properties | Back Directory | [Appearance]
white to light yellow crystal powder | [Melting point ]
97-100 °C (lit.) | [Boiling point ]
303.85°C (rough estimate) | [density ]
1.4251 (rough estimate) | [refractive index ]
1.6404 (estimate) | [storage temp. ]
0-6°C
| [solubility ]
Chloroform (Slightly), DMSO (Slightly), Methanol (Slightly) | [form ]
Powder | [color ]
Yellow | [Water Solubility ]
Insoluble in water. | [BRN ]
1869747 | [InChI]
InChI=1S/C14H9Br/c15-14-12-7-3-1-5-10(12)9-11-6-2-4-8-13(11)14/h1-9H | [InChIKey]
ZIRVQSRSPDUEOJ-UHFFFAOYSA-N | [SMILES]
C1=C2C(C=C3C(=C2Br)C=CC=C3)=CC=C1 | [CAS DataBase Reference]
1564-64-3(CAS DataBase Reference) | [NIST Chemistry Reference]
9-Bromoanthracene(1564-64-3) |
| Safety Data | Back Directory | [Hazard Codes ]
Xi | [Safety Statements ]
S22:Do not breathe dust .
S24/25:Avoid contact with skin and eyes . | [WGK Germany ]
3
| [Hazard Note ]
Irritant | [HS Code ]
29039900 |
| Questions And Answer(Q&A) | Back Directory | [Description]
9-bromoanthracene is a kind of bromine-derived anthracene. It is known to be able to reversibly photodimerize in a head-to tail fashion upon irradiation by long-wavelength ultraviolet light. The photodimers of 9-bromoanthracene are suitable to be used as alkyl halide initiators in the atom transfer radical polymerization (ATRP) reactions. It is also used as the intermediate for the preparation of the 9-substitued form of the polycyclic aromatic hydrocarbon (PAH) anthracene.
| [Preparation]
Anthracene (5 g, 28.05 mmol) was dissolved in CHCl3. Then N-bromosuccinimide (NBS,4.99 g, 28.05 mmol) was added in batches away from light, and the reaction solution was continuously stirred for 12 h. The resulting mixture was stirred for another 30 min with appropriate water, and extracted with CH2Cl2. The CH2Cl2 solution was dried over anhydrous MgSO4. After removing CH2Cl2 solvent, the residue was recrystallized from anhydrous ethanol to give 4.78 g (66.3 %) of a green-yellow needle solid. 1H NMR (500 MHz, CDCl3) δ 8.55 (d, J = 8.9 Hz, 2H), 8.48 (s, 1H), 8.03 (d, J = 8.4 Hz, 2H), 7.67 – 7.60 (m, 2H), 7.56 – 7.51 (m, 2H). EI-MS (m/z): Calculated for C14H9Br: 257.13. Found [M+ ]: 255.96.
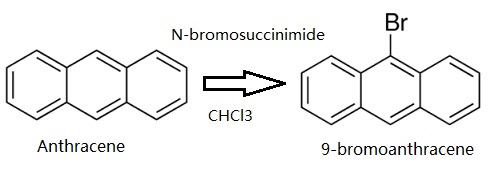
Synthesis of 9-bromoanthracene
| [References]
Cohen, Nicole A., et al. Macromolecular Chemistry and Physics 210.3 ‐4 (2009): 263-268.
Xu, Xiaoming, Wenzhe Lu, and Richard B. Cole. Analytical chemistry 68.23 (1996): 4244-4253.
Dang, Hung, and Miguel A. Garcia-Garibay. Journal of the American Chemical Society 123.2 (2001): 355-356.
|
| Hazard Information | Back Directory | [Chemical Properties]
white to light yellow crystal powder | [Uses]
9-Bromoanthracene acts as an intermediate in the preparation of 9-substituted derivative of the polycyclic aromatic hydrocarbon (PAH) anthracene. | [Synthesis Reference(s)]
The Journal of Organic Chemistry, 57, p. 2740, 1992 DOI: 10.1021/jo00035a038 | [Purification Methods]
Crystallise 9-bromoanthracene from MeOH or EtOH followed by sublimation in vacuo. [Masnori et al. J Am Chem Soc 108 126 1986, Beilstein 5 IV 2295.] |
|
|