| Identification | Back Directory | [Name]
OCTAETHYLENEGLYCOL OCTYL ETHER | [CAS]
26468-86-0 | [Synonyms]
EH 6
PEH 6
JFC-35
Berol 840
Newcol 1004
Newcol 1006
Newcol 1008
Newcol 1020
Koremul OT 5
Blaunon EH 2
Blaunon EH 4
Blaunon EH 6
Ecosurf EH 6
Ethylan EH 4
Newcol 1008GC
Nonion EH 202
Nonion EH 203
ralufonen16-80
Imbentin OA 100
Nissan Nonion EH 202
Nissan Nonion EH 203
RALUFON (R) EN 16-80
Ethylhexanol?ethoxylate
2-ethylhexanol ethoxylate
Ethoxylated 2-ethylhexanol
OCTAETHYLENEGLYCOL OCTYL ETHER
Polyethyleneglycol 2-ethylhexyl ether
Polyethyleneglycolmono(2-ethylhexyl)ether
OCTAETHYLENEGLYCOL OCTYL ETHER ISO 9001:2015 REACH
α-(2-Ethylhexyl)-ω-hydroxy-poly(oxy-1,2-ethanediyl)
Poly(oxy-1,2-ethanediyl),a-(2-ethylhexyl)-w-hydroxy-
alpha-(2-ethylhexyl)-omega-hydroxy-poly(oxy-2-ethanediyl)
2-ethanediyl),.alpha.-(2-ethylhexyl)-.omega.-hydroxy-Poly(oxy-1
Poly(oxy-1,2-ethanediyl),.alpha.-(2-ethylhexyl)-.omega.-hydroxy-
Polyethylenglykol, Mono(2-ethylhexyl)ether mit 4-35 Mol Ethylenoxid | [EINECS(EC#)]
607-943-2 | [Molecular Formula]
C24H50O9 | [MDL Number]
MFCD00804449 | [MOL File]
26468-86-0.mol | [Molecular Weight]
482.648 |
| Hazard Information | Back Directory | [Chemical Properties]
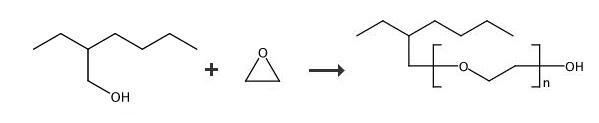
Polyethylene glycol mono(2-ethylhexyl) ether is a low foaming nonionic surfactant with a high cloud point and used as a brightener in acid zinc baths. | [Uses]
Use as wetting agent, permeating agent. For example, used in textile, leather, chemical fiber.
Use as cleansing agent. For example, used in personal care products, household cleaning, industrial cleaning, textile, leather, chemical fiber.
Use as emulsifying agent, dispersing agent.
Use as solubilizing agent. | [Synthesis]
Preparation of Polyethylene glycol mono(2-ethylhexyl) ether: In a first stage, a 1003g sample of 2-ethylhexanol (EH) was charged to a one-gallon alkoxylation reactor and dehydrated at 115-120°C under a slight nitrogen purge until the moisture content was less than 0.05%. After the dehydrated alcohol was cooled to about 90°C, 3g of boron trifluoride-etherate, included as a catalyst, was charged to the reactor. The alcohol- catalyst mixture was then mixed for 10 minutes, then purged with nitrogen 5 times to remove air and heated to 105°C. Next, 684g of ethylene oxide (EO) was added to the reactor. An exothermic reaction occurred immediately and cooling was applied throughout the EO addition to maintain the reaction temperature within the range of 105-115°C. Following the EO addition, the reaction mixture was digested at 115°C for one hour then purged with nitrogen five times and cooled to 90°C. In a second stage, 31 g of 45% potassium hydroxide, included as a second catalyst, was charged to the reactor. The mixture was then mixed for 10 minutes and then dehydrated at 110°C under nitrogen purge. When the moisture content of the mixture was less than 0.1% (after about 120 minutes of dehydration) 665g of EO was then added to the reactor. Here again, an exothermic reaction occurred and cooling was applied throughout the EO addition to maintain the reaction temperature within the range of 120-130°C. Following the EO addition, the ethoxylation reaction mixture was digested at 125°C-130°C for one hour, then purged with nitrogen five times and cooled to 65°C. Next, 16g of acetic acid was added and then the product mixture was mixed for 15 minutes then discharged. About 2270g of alcohol ethoxylate was collected.
 |
|
|