| Identification | More | [Name]
Dichlorodimethylsilane | [CAS]
75-78-5 | [Synonyms]
DICHLORODIMETHYLSILANE
DICHLORODIMETHYLSILICANE
DIMETHYLDICHLOROSILANE
DIMETHYLSILICON DICHLORIDE
DMDCS
DOW CORNING(TM) PRODUCT Z-1219
DOW CORNING Z-1219 CHLOROSILANE
FLUKA GEL REPEL I
FLUKA GEL REPEL II
GLASS-PREP(TM)
SILANE M2
SILANISATION SOLUTION
SILANIZATION SOLUTION I
SILANIZATION SOLUTION II
(CH3)2SiCl2
dichlorodimethyl-silan
Dichlorodimethylsilicon
dimethyldichlorosilicane
Dimethyl-dichlorsilan
dimethyl-dichlorsilan(czech) | [EINECS(EC#)]
200-901-0 | [Molecular Formula]
C2H6Cl2Si | [MDL Number]
MFCD00000491 | [Molecular Weight]
129.06 | [MOL File]
75-78-5.mol |
| Chemical Properties | Back Directory | [Appearance]
Colorless to brown liquid | [Melting point ]
-76 °C | [Boiling point ]
70 °C(lit.)
| [density ]
1.333 g/mL at 20 °C
| [vapor pressure ]
<200 hPa (20 °C) | [refractive index ]
n20/D 1.500
| [Fp ]
3 °F
| [storage temp. ]
Store at 0-5°C | [solubility ]
sol chlorinated solvents and ethereal solvents; reacts
with protic solvents. | [form ]
liquid | [color ]
colorless | [Specific Gravity]
1.0637 | [Stability:]
Stable. Reacts violently with water and alcohols. Highly flammable. Incompatible with strong oxidizing agents, water, alcohols, caustics, ammonia. | [explosive limit]
1.75-48.5%(V) | [Water Solubility ]
reacts | [Hydrolytic Sensitivity]
8: reacts rapidly with moisture, water, protic solvents | [Sensitive ]
Moisture Sensitive | [Detection Methods]
GC,NMR | [BRN ]
605287 | [CAS DataBase Reference]
75-78-5(CAS DataBase Reference) | [NIST Chemistry Reference]
Silane, dichlorodimethyl-(75-78-5) | [Storage Precautions]
Store under nitrogen;Moisture sensitive | [EPA Substance Registry System]
75-78-5(EPA Substance) |
| Safety Data | Back Directory | [Hazard Codes ]
Xn,Xi,F,N | [Risk Statements ]
R20:Harmful by inhalation.
R59:Dangerous for the ozone layer.
R36/37/38:Irritating to eyes, respiratory system and skin .
R11:Highly Flammable.
R67:Vapors may cause drowsiness and dizziness.
R65:Harmful: May cause lung damage if swallowed.
R63:Possible risk of harm to the unborn child.
R48/20:Harmful: danger of serious damage to health by prolonged exposure through inhalation .
R38:Irritating to the skin.
R20/21:Harmful by inhalation and in contact with skin .
R50/53:Very Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment . | [Safety Statements ]
S60:This material and/or its container must be disposed of as hazardous waste .
S61:Avoid release to the environment. Refer to special instructions safety data sheet .
S62:If swallowed, do not induce vomiting: seek medical advice immediately and show this container or label .
S36/37:Wear suitable protective clothing and gloves .
S59:Refer to manufacturer/supplier for information on recovery/recycling . | [RIDADR ]
UN 2924 3/PG 2
| [WGK Germany ]
3
| [RTECS ]
VV3150000
| [F ]
3-10-19-21 | [Autoignition Temperature]
425 °C | [TSCA ]
Yes | [HazardClass ]
3 | [PackingGroup ]
II | [HS Code ]
29310095 | [Safety Profile]
Poison by ingestion and
intraperitoneal routes. Moderately toxic by
inhalation. A skin and severe eye irritant.
Violent reaction on contact with water.
When heated to decomposition it emits
toxic fumes of Cl-. See also
CHLOROSILANES. | [Hazardous Substances Data]
75-78-5(Hazardous Substances Data) | [Toxicity]
LD50 orally in Rabbit: 6056 mg/kg |
| Raw materials And Preparation Products | Back Directory | [Raw materials]
Copper(I) chloride-->Chlorotrimethylsilane-->Chloromethane-->Silicon-->Methyltrichlorosilane | [Preparation Products]
Silicon carbide-->Chlorodimethylphenylsilane-->Octamethylcyclotetrasiloxane-->tert-Butyldimethylsilyl chloride-->Silk softener L-->Xanthophyll-->1,1,3,3,5,5-hexamethyl-1,3,5-trisilacyclohexane-->SILICONE RESIN-->Silica, Hydrophobic-->Dicyanodimethylsilane-->POLYDIMETHYLSILOXANE-->PolydimettIylsiloxane-->hydroxyl silicone oil-203-->Chlorodimethylsilane-->Dimethyldimethoxysilane-->3-TRIMETHYLSILYL-2-PROPYN-1-OL-->Chloromethyldichloromethylsilane-->Hexamethylcyclotrisiloxane-->1,1,3,3-tetramethyldisiloxane-1,3-diol |
| Hazard Information | Back Directory | [General Description]
A colorless fuming liquid with a pungent odor. Flash point 16°F. Vapor and liquid may cause burns. Denser than water. Vapors heavier than air. | [Reactivity Profile]
Chlorosilanes, such as DIMETHYLDICHLOROSILANE, are compounds in which silicon is bonded to from one to four chlorine atoms with other bonds to hydrogen and/or alkyl groups. Chlorosilanes react with water, moist air, or steam to produce heat and toxic, corrosive fumes of hydrogen chloride. They may also produce flammable gaseous H2. They can serve as chlorination agents. Chlorosilanes react vigorously with both organic and inorganic acids and with bases to generate toxic or flammable gases. | [Health Hazard]
Inhalation irritates mucous membranes. Severe gastrointestinal damage may occur. Vapors cause severe eye and lung injury. Upon short contact, second and third degree burns may occur. | [Fire Hazard]
Vapor may explode if ignited in an enclosed area. Reacts vigorously with water to generate hydrogen chloride. Hydrogen chloride and phosgene gases may be formed upon heating or in fire. Runoff to sewer may create fire or explosion hazard. | [Chemical Properties]
Colorless to brown liquid | [Physical properties]
mp ?76°C; bp 70–71°C; d 1.064 g cm?3. | [Uses]
Dichlorodimethylsilane can be used as additive for pinacol cyclization; protecting group for diols and
carbonyl compounds;precursor for a wide variety of siliconbased
reagents.
Dichlorodimethylsilane (1) allows clean
pinacol cyclization of a keto aldehyde to occur without competition
from an aldol reaction (eq 1).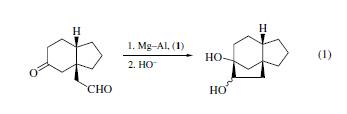
| [Uses]
Dichlorodimethylsilicon is a organosilicon compound and is the precursor to dimethylsilicone and polysilane compounds. | [Production Methods]
Produced by the action of silicon on methyl chloride in
presence of copper catalyst, or by Grignard reaction from
methyl chloride and silicon tetrachloride. | [reaction suitability]
reagent type: derivatization reagent
reaction type: Silylations | [Potential Exposure]
PrimaryIrritant. Used as an intermediate in the manufacture of silicone polymers. | [First aid]
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting. Medical observation isrecommended for 2448 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray. | [storage]
(1) Color Code—Red: Flammability Hazard: Storein a flammable liquid storage area or approved cabinetaway from ignition sources and corrosive and reactivematerials. (2) Color Code—Blue: Health Hazard/Poison:Store in a secure poison location. Dimethyldichlorosilanemust be stored to avoid contact with oxidizers (such as perchlorates, peroxides, permanganates, chlorates, andnitrates), since violent reactions occur. Before entering confined space where this chemical may be present, check tomake sure that an explosive concentration does not exist.Store in tightly closed containers in a cool, well-ventilatedarea away from water, steam, or moisture, because toxicand corrosive hydrogen chloride gas can be produced. Donot store at temperatures above 50℃/122°F. Sources ofignition, such as smoking and open flames, are prohibitedwhere dimethyldichlorosilane is handled, used, or stored.Metal containers involving the transfer of 5 gallons or moreof dimethyldichlorosilane should be grounded and bonded. | [Shipping]
Dimethyldichlorosilane requires a shipping labelof “FLAMMABLE LIQUID, CORROSIVE.” It falls inHazard Class 3 and Packing Group II | [Purification Methods]
Other impurities are chlorinated silanes and methylsilanes. Fractionate it through a 3/8in diameter 7ft Stedman column (p 11) rated at 100 theoretical plates at almost total reflux. See purification of MeSiCl2. Solutions in heptane, 1,1,1-trichloroethane or 1-chloronaphthalene are used for the silanization of glassware and pipettes. [Sauer & Hadsell J Am Chem Soc 70 3590 1948, Beilstein 4 IV 4110.] | [Incompatibilities]
Forms explosive gas mixture with air.Water, steam, and moisture forms toxic and corrosivehydrogen chloride gas. Incompatible with acetone, amines,ammonia, alcohols, strong oxidizers, caustics. Attacks mostmetals. Do not store in temperatures above 122°F/50℃. | [Toxics Screening Level]
The chronic initial threshold screening level (ITSL) for dimethyl dichlorosilane is 1 μg/m3 (annual averaging time) based on the Michigan Department of Environmental Quality (MDEQ), AQD Rule 336.1232 (1) (d). |
| Questions And Answer | Back Directory | [Description]
Dichlorodimethylsilane is a tetrahedral, organosilicon compound with the molecular formula Si(CH3)2Cl2. The compound is a colorless liquid at room temperature, has a pungent odor and readily reacts with water to form both cyclic and linear Si-O chains. On an industrial scale, dichlorodimethylsilane is manufactured as the main precursor to polysilane and dimethylsilicone compounds.
| [History]
James Crafts and Charles Friedel are American chemists who first reported organosilicon compounds in 1863 by synthesizing tetraethylsilane from silicon tetrachloride and diethylzinc. Nevertheless, major progress in organosilicon chemistry occurred when Fredrick Kipping and his students reacted tetrachloride with Grignard reagents to produce diorganodichlorosilanes (R2SiCl2), which they used for their experiments. The dpemand for silicones increased in the 1930s as many aircraft companies needed better insulators for sealing materials and electric motors for aircraft engines; therefore, there was need for efficient synthesis of dichlorodimethylsilane.
| [Preparation]
Dichlorodimethylsilane is prepared by passing methyl chloride through a heated tube packed with copper (I) chloride and ground silicon using Cu2O as the catalyst. Methyl chloride is the passed through a reactor to produce dichlorodimethylsilane.
2 CH3Cl + Si → (CH3)2SiCl2
The other products in this reaction apart from dichlorodimethylsilane include CH3)3SiCl, CH3SiHCl2, and CH3SiCl3, which can be separated fromone another by fractional distillation.
| [Applications]
Dichlorodimethylsilane is majorly used in the production of silicones. Moreover, it is utilized in the synthesis of polysilanes, which are the main precursors to silicon carbide. Dichlorodimethylsilane can be used to coat glass to prevent the adsorption of micro-particles.
|
|
|