[[difluoro(triMethylsilyl)Methyl]thio]-Benzene
|
|
|
- CAS-Nr.
- 536975-49-2
- Englisch Name:
- [[difluoro(triMethylsilyl)Methyl]thio]-Benzene
- Synonyma:
- [Difluoro(phenylthio)Methyl]triMethylsilane;[[difluoro(triMethylsilyl)Methyl]thio]-Benzene;[Difluoro(phenylsulfanyl)methyl]trimethylsilane;Benzene, [[difluoro(trimethylsilyl)methyl]thio]-;[Difluoro(phenylsulfanyl)methyl]trimethylsilane - [D6812]
- CBNumber:
- CB52644988
- Summenformel:
- C10H14F2SSi
- Molgewicht:
- 232.37
- MOL-Datei:
- 536975-49-2.mol
|
[[difluoro(triMethylsilyl)Methyl]thio]-Benzene Eigenschaften
- Löslichkeit
- soluble in most organic solvents, for example hexane,
tetrahydrofuran (THF), CH2Cl2, dimethylformamide (DMF),
and so on.
Sicherheit
- Risiko- und Sicherheitserklärung
- Gefahreninformationscode (GHS)
| Bildanzeige (GHS) |

|
| Alarmwort |
Warnung
|
| Gefahrenhinweise |
| Code |
Gefahrenhinweise |
Gefahrenklasse |
Abteilung |
Alarmwort |
Symbol |
P-Code |
| H302 |
Gesundheitsschädlich bei Verschlucken. |
Akute Toxizität oral |
Kategorie 4 |
Warnung |
 |
P264, P270, P301+P312, P330, P501 |
| H315 |
Verursacht Hautreizungen. |
Hautreizung |
Kategorie 2 |
Warnung |
 |
P264, P280, P302+P352, P321,P332+P313, P362 |
| H319 |
Verursacht schwere Augenreizung. |
Schwere Augenreizung |
Kategorie 2 |
Warnung |
 |
P264, P280, P305+P351+P338,P337+P313P |
| H335 |
Kann die Atemwege reizen. |
Spezifische Zielorgan-Toxizität (einmalige Exposition) |
Kategorie 3 (Atemwegsreizung) |
Warnung |
 |
|
|
| Sicherheit |
| P280 |
Schutzhandschuhe/Schutzkleidung/Augenschutz tragen. |
| P305+P351+P338 |
BEI KONTAKT MIT DEN AUGEN: Einige Minuten lang behutsam mit Wasser spülen. Eventuell vorhandene Kontaktlinsen nach Möglichkeit entfernen. Weiter spülen. |
|
[[difluoro(triMethylsilyl)Methyl]thio]-Benzene Chemische Eigenschaften,Einsatz,Produktion Methoden
Beschreibung
An effective reagent to introduce difluoromethyl groups into carbonyls, imines, enamines, and
alkyl halides. Not only various simple aldehydes and ketones, but also functionalized carbonyls
such as α- and γ-ketoesters and cyclic imides can be difluoro(phenylthio)methylated in high yields
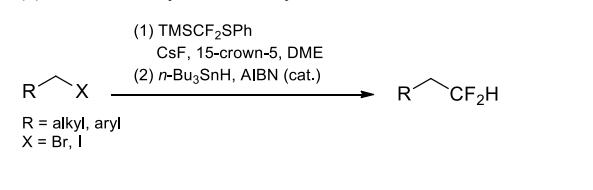
under the activation of a catalytic amount of Lewis bases. The substitution reaction proceeds well
with primary alkyl bromides and iodides as the limiting reactant when cesium
fluorode/15-crown-5 is used as the fluoride source/additive. Under radical conditions, the
difluoro(phenylthio)methyl compounds containing vinyl functional groups can form 5- or
6-membered rings via intramolecular cyclization.
Physikalische Eigenschaften
colorless liquid; bp 86–87C/4 mmHg.
Verwenden
Organofluorine compounds have received remarkable
interest in recent years due to their wide-ranging biological
effects. The development of general synthetic routes to such compounds
and the use of new fluorinated compounds as building
blocks are of great importance. Of particular interest is the selective
incorporation of the gem-difluoromethylene group ‘CF2’ into
organic molecules. The report on the synthesis of PhSCF2SiMe3
was published in 2003 by Prakash et al., and the reagent has
become one of the most versatile and efficient nucleophilic
(phenylthio)difluoromethylating reagents.
When excess potassium t-butoxide
was used as a promoter, PhSCF2SiMe3 reacted with diphenyl
disulfide to give the corresponding dithioacetal in 85%
yield (eq 8).
synthetische
[difluoro(phenylthio)methyl]trimethylsilane
(PhSCF2SiMe3) was prepared for the first time by
Prakash et al.,1 using the Barbier coupling reaction of bromodifluoromethylphenyl
sulfide,2 prepared from dibromodifluoromethane
and sodium benzenethiolate,3 magnesium metal, and
chlorotrimethylsilane (TMSCl) in DMF (eq 1).
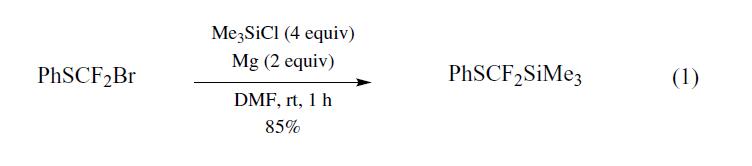
Reaktionen
(1) Difluoromethylation of aldehydes and ketones.

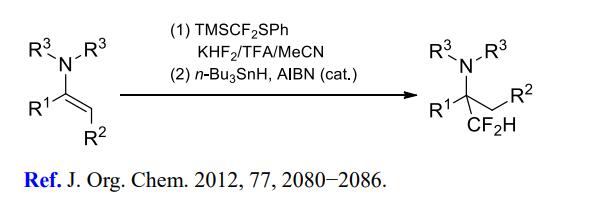
(2) Difluoromethylation of imines and enamines.
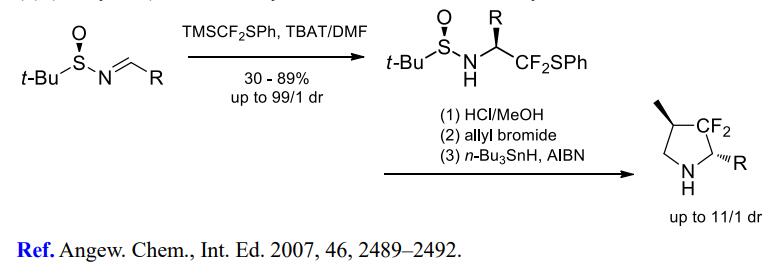
(3) (Phenylthio)difluoromethylation of imines for further cyclizations.
(4) Difluoromethylation of alkyl halides.
[[difluoro(triMethylsilyl)Methyl]thio]-Benzene Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte
[[difluoro(triMethylsilyl)Methyl]thio]-Benzene Anbieter Lieferant Produzent Hersteller Vertrieb Händler.
Global( 19)Lieferanten
- [[difluoro(triMethylsilyl)Methyl]thio]-Benzene
- [Difluoro(phenylthio)Methyl]triMethylsilane
- [Difluoro(phenylsulfanyl)methyl]trimethylsilane
- Benzene, [[difluoro(trimethylsilyl)methyl]thio]-
- [Difluoro(phenylsulfanyl)methyl]trimethylsilane - [D6812]
- 536975-49-2
- C10H14F2SSi

