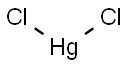Mercuric Chloride
- CAS No.
- 7487-94-7
- Chemical Name:
- Mercuric Chloride
- Synonyms
- HgCl2;MERCURIC CHLORIDE;Mercury dichloride;MERCURY(II) CHLORIDE;MERCURIC BICHLORIDE;LHGG;Sulem;tl898;TL 898;Sulema
- CBNumber:
- CB00126259
- Molecular Formula:
- Cl2Hg
Lewis structure

- Molecular Weight:
- 271.5
- MDL Number:
- MFCD00011041
- MOL File:
- 7487-94-7.mol
| Melting point | 277 °C(lit.) |
|---|---|
| Boiling point | 302 °C |
| Density | 5.44 |
| bulk density | 2000kg/m3 |
| vapor pressure | 1.3 mm Hg ( 236 °C) |
| refractive index | 1.859 |
| Flash point | 302°C |
| storage temp. | Store at RT. |
| solubility | H2O: soluble |
| form | powder |
| color | White |
| Specific Gravity | 5.44 |
| PH | 3.2 (15g/L in H2O) |
| Odor | odorless |
| Water Solubility | 7.4 g/100 mL (20 ºC) |
| Merck | 14,5876 |
| Dielectric constant | 3.2(Ambient) |
| Stability | Stable, but moisture sensitive and light sensitive - decomposes in sunlight. Incompatible with strong acids, ammonia, carbonates, metallic salts, alkalies, phosphites, phosphates, sulfites, sulfates, arsenic, antimony, bromides. |
| CAS DataBase Reference | 7487-94-7(CAS DataBase Reference) |
| FDA 21 CFR | 310.545 |
| EWG's Food Scores | 4 |
| FDA UNII | 53GH7MZT1R |
| ATC code | D08AK03 |
| NIST Chemistry Reference | Mercury dichloride(7487-94-7) |
| EPA Substance Registry System | Mercuric chloride (7487-94-7) |
| UNSPSC Code | 12352302 |
| NACRES | NA.21 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |     GHS05,GHS06,GHS08,GHS09 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H300-H314-H341-H361f-H372-H410 | |||||||||
| Precautionary statements | P260-P273-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | T+,N,T | |||||||||
| Risk Statements | 28-34-48/24/25-50/53-51/53-48/21/22-25-68-62-27-24/25 | |||||||||
| Safety Statements | 36/37/39-45-60-61-28-26-36/37 | |||||||||
| RIDADR | UN 1624 6.1/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | OV9100000 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 6.1 | |||||||||
| PackingGroup | II | |||||||||
| Hazardous Substances Data | 7487-94-7(Hazardous Substances Data) | |||||||||
| NFPA 704 |
|
Mercuric Chloride Chemical Properties,Uses,Production
Description
Mercury is found in trace amounts in the air, but the largest
deposits on earth are as cinnabar (mercuric sulfide). It exists in
several forms such as a liquid metal (quicksilver), as a vapor,
and in compounds (organic and inorganic). Mercury has been
used as a medicine, to make amalgams, and in many industrial
applications. Exposure to mercury can be toxic in any of its
forms, in any route (ingestion, skin contact, and inhalation)
depending on the dose and duration of exposure. Thermometers,
‘button’ batteries, the new energy-saving fluorescent light
bulbs, and many seafood (shellfish, tuna, marlin, and many
others) contain mercury, and are, therefore, potential sources
of mercury poisoning. The primary intracellular target of
mercury is the sulfhydryl groups in many tissue enzymes and
proteins. Binding of –SH– groups paralyzes vital functions,
eventually causing failure of organ systems such as the lungs,
kidneys, or the nervous system.
Industrial disasters cause release of mercury or methylmercury
into the environment. The classic example of such
a disaster is the contamination of Minamata Bay in Japan,
where the term Minamata disease originated. Studies from
about 1956 to 1960 suggested the unusual symptoms
(neurological) found in people in this area was traced back to
industrial wastewater containing methylmercury. Nearly 2200
people were diagnosed and over 1700 deaths were eventually
attributed to methylmercury poisoning. Mercury has been used
in skin creams; the most recent problem cream was identified
in 1996 from Mexico named ‘Crèma de Belleza-Manning.’
Mercury poisoning can be caused by all forms of mercury
(elemental, vapor, inorganic, and organic).
Chemical Properties
Mercury bichloride is an odorless white crystalline solid.
Chemical Properties
Mercuric chloride,HgC12, is white crystals that are soluble in water and alcohol that melt at 276℃ and boil at 302℃. Highly toxic and corrosive, it is used in the manufacture of mercury compounds, in organic synthesis, as a reagent and catalyst, as a fungicide, insecticide, and wood preservative, and for many other purposes.
Uses
Mercuric chloride is used in preservatives for wood and anatomical specimens, embalming solutions, disinfectants, photographic intensifiers, leather tanning, seed treatments, analytical reagents for organic syntheses, and the manufacture of other mercury-containing compounds. Pharmaceuticals containing mercuric chloride have also been used therapeutically as topical antiseptics and disinfectants.
Uses
Mercuric chloride is used in tanning leather; intensifier in photography; topical antiseptic and disinfectant.
Uses
Preserving (kyanizing) wood and anatomical specimens; also embalming; disinfecting; browning and etching steel and iron; intensifier in photography; white reserve in fabric printing; tanning leather; electroplating aluminum; depolarizer for dry batteries; freeing gold from lead; magic photograms; mordant for rabbit and beaver furs; staining wood and vegetable ivory pink; manufacture of ink for mercurography; treating seed potatoes; manufacture of other mercury Compounds. As an important reagent In animal chemistry.
Definition
ChEBI: A mercury coordination entity made up of linear triatomic molecules in which a mercury atom is bonded to two chlorines. Water-soluble, it is highly toxic. Once used in a wide variety of applications, including preserving wood and anatomical specimens, emba ming and disinfecting, as an intensifier in photography, as a mordant for rabbit and beaver furs, and freeing gold from lead, its use has markedly declined as less toxic alternatives have been developed.
General Description
An odorless white crystalline solid. Density 5.4 g / cm3. Melting point 277°C. Slightly volatile at ordinary temperatures. Can be sublimed unchanged. Corrosive to the mucous membranes. Toxic by inhalation (dusts, etc.), ingestion, and skin absorption. Used in photography, disinfectants, wood preservatives, fungicides.
Air & Water Reactions
Slightly soluble in water.
Reactivity Profile
Mercury chloride is decomposed by sunlight. Incompatible with formates sulfites, phpophosphites, phosphates, sulfides, albumin, gelatin, alkalis, alkaloid salts, ammonia, lime water, antimony, arsenic, bromides, borax, carbonates, reduced iron, iron, copper, lead and silver salts, infusions of cinchona, oak bark or senna, tannic acids and vegetable astringents. .
Hazard
Toxic by ingestion, inhalation, and skin absorption; a poison.
Health Hazard
Mercury chloride is classified as extremely toxic. All forms of mercury are poisonous if absorbed. Probable oral lethal dose is 5-50 mg/kg; between 7 drops and 1 teaspoonful for a 150 lb. person. Mercury chloride is one of the most toxic salts of mercury. Material attacks the gastrointestinal tract and renal systems.
Fire Hazard
Material may explode on heating, with friction, or contact with alkali metals, sulfides, acetylene, ammonia, and oxalic acid. Upon decomposition highly toxic chloride and mercury fumes are emitted. Avoid formates, sulfites, hypophosphites, phosphates, sulfides, albumin, gelatin, alkalies, alkaloid salts, ammonia, lime water, antimony, arsenic, bromides, borax, carbonates, reduced iron, copper, iron, lead, silver salts, infusions of cinchona, columbo, oak bark or senna, and tannic acid. Mercury chloride may explode with friction or application of heat. Mixtures of Mercury chloride and sodium or potassium are shock sensitive and will explode on impact. Avoid contact with acids or acid fumes.
Safety Profile
A human poison by ingestion. Poison experimentally by ingestion, skin contact, and subcutaneous routes. Human systemic effects by ingestion: respiratory obstruction, nausea or vomiting, urine volume decrease or anuria. Human reproductive effects by ingestion: terminates pregnancy. Experimental teratogenic and reproductive effects. Human mutation data reported. Questionable carcinogen. A severe eye and skin irritant. Reaction with sodmm aci-nitromethanide + acids forms the explosive mercury fulminate. Reacts violently with K, Na. When heated to decomposition it emits toxic fumes of Hg.
Potential Exposure
Mercuric chloride is used as dip for bulbs and tubers; for earthworm control; as repellent to ants, roaches, etc.; in preserving wood and anatomical specimens; embalming, browning, etching steel and iron; as a catalyst for organic synthesis; disinfectant, antiseptic, tanning; textile printing aid; manufacture of dyes; in agricultural chemicals; dry batteries; pharmaceuticals, and photographic chemicals
First aid
If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least 15 min, occasionally lifting upper and lower lids. Seek 1686 Mercuric chloride medical attention immediately. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart action has stopped. Transfer promptly to a medical facility. When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit. Medical observation is recommended for 2448 h after breathing overexposure, as pulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consider administering a corticosteroid spray. Antidotes and special procedures for medical personnel: The drug NAP (n-acetyl penicillamine) has been used to treat mercury poisoning, with limited success. Note to physician: For severe poisoning BAL [British AntiLewisite, dimercaprol, dithiopropanol (C3H8OS2)] has been used to treat toxic symptoms of certain heavy metals poisoning including mercury. Although BAL is reported to have a large margin of safety, caution must be exercised, because toxic effects may be caused by excessive dosage. Most can be prevented by premedication with 1-ephedrine sulfate (CAS: 134-72-5).
Environmental Fate
Mercury adsorbed from mercuric chloride and 2-methoxyethylmercury chloride (Aretan) solutions by three contrasting soils showed a dependence on soil–solution ratio and initial mercury (Hg) concentration in soil solution. Changing the soil–solution ratio from 1:10 to 1:100 but keeping the initial concentration constant resulted in an increase in initial concentration but, on the other hand, resulted in decrease in Hg adsorption. Upon manipulation of the pH of the surface soils, adsorption of mercuric chloride at 100 mg Hg l-1 concentration increased from ~ 70 to over 95 mg Hg kg-1 when the pH was raised from 5.0 to 8.0. Precipitation of Hg may also have contributed to this trend. Removal of organic matter from soil resulted in large reductions of Hg adsorbed, as much as 95% from the mercuric chloride solutions. Mercuric compounds found in the atmosphere are likely to be transformed by chemical or physical processes. Theoretical calculations on the photodissociation of mercuric compounds have indicated that mercuric chloride and mercuric cyanide are stable, while mercuric hydroxide may dissociate in the gas phase. Exchange reactions between water and mercury compounds are likely to occur in the atmosphere. These exchange reactions eventually result in the release of elemental mercury into the gaseous phase.
storage
Color Code—Blue: Health Hazard/Poison: Store in a secure poison location. Prior to working with this chemical you should be trained on its proper handling and storage. Mercuric chloride must be stored to avoid contact with potassium and sodium, since violent reactions occur. See also “Incompatibilities,” above. Store in tightly closed containers in a cool, well-ventilated area.
Shipping
UN1624 Mercuric chloride, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Purification Methods
It is soluble in EtOH and is extracted into Et2O from an aqueous solution. It is very POISONOUS and 0.2-0.4g is fatal. The antidote is immediate administration of white of egg as an emetic.
Toxicity evaluation
A reference from Middle Ages in Goldwater’s book on mercury describes oral ingestion of mercury as causing severe abdominal cramps, bloody diarrhea, and suppression of urine. This is an accurate report of the effects following accidental or suicidal ingestion of mercuric chloride. Injection of mercuric chloride produces necrosis of the epithelium of the pars recta kidney. Cellular changes include fragmentation and disruption of the plasma membrane and its appendages, vesiculation and disruption of the endoplasmic reticulum and other cytoplasmic membranes, dissociation of polysomes and loss of ribosomes, mitochondrial swelling and loss of amorphous intramatrical deposits, and condensation of nuclear chromatin. Although exposure to a high dose of mercuric chloride is directly toxic to renal tubular lining cells, chronic low-dose exposure may induce an immunologic glomerular disease. This form of chronic mercury injury to the kidney is clinically the most common form of mercury-induced nephropathy. Experimental studies have shown that the pathogenesis of chronic mercury nephropathy has two phases: an early phase characterized by anti-basement membrane glomerular nephritis followed by a superimposed immune complex glomerulonephritis with transiently raised concentrations of circulating immune complexes.
Incompatibilities
A strong reducing agent; keep away from oxidizers. Mercuric chloride may explode with friction or application on heat. Mixtures of mercuric chloride and sodium or potassium are shock sensitive and will explode on impact. Avoid contact with acids or acid fumes. Also avoid the presence of formats, sulfites, hypophosphites, phosphates, sulfide; albumin, gelatin, alkalies, alkaloid salts; ammonia, lime water; antimony, arsenic, bromides, borax, carbonates, reduced iron, copper; iron, lead, silver salts; infusions of cinchona; columbo, oak bark or senna; and tannic acid
Mercuric Chloride Preparation Products And Raw materials
Raw materials
Preparation Products
1of2