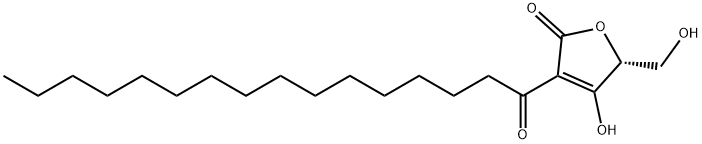
(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone
- CAS No.
- 150627-37-5
- Chemical Name:
- (R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone
- Synonyms
- RK-682 >=98% (HPLC);(R)-3-Hexadecanoyl-5-hydroxymethyltetronic acid;(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone;2(5H)-Furanone, 4-hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-, (5R)-
- CBNumber:
- CB02750381
- Molecular Formula:
- C21H36O5
- Molecular Weight:
- 368.51
- MDL Number:
- MFCD20488046
- MOL File:
- 150627-37-5.mol
| Melting point | 105-107 °C |
|---|---|
| Boiling point | 546.7±50.0 °C(Predicted) |
| Density | 1.073±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | DMSO, heptane and xylene: ≥8mg/mL |
| pka | 1.80±0.10(Predicted) |
| form | powder |
| color | white to tan |
| Stability | Stable for 1 year from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20° for up to 3 months. |
(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone price More Price(2)
(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone Chemical Properties,Uses,Production
Description
(±)-RK-682 (150627-37-5) is a protein tyrosine phosphatase inhibitor (IC50‘s = 54 7 μM for CD45, 2.0 μM for VHR; did not inhibit cdc25B) originally isolated from the fermentation of Streptomyces sp. 88-682.1?Inhibits cell cycle at G1/S. RK-682 has also been shown to inhibit PLA2 (IC50?= 16 μM)2, HIV-1 protease (IC50?= 84 μlM)3, and heparanase (IC50?= 17 μM)4. Natural RK-682 (R-isomer) and synthetic racemic material have identical phosphatase activity.5?Care should be taken when using RK-682 in the presence of metal salts – because it readily forms metal complexes that affects its phosphatase inhibitory activity.6?RK-682 has been identified as a potential promiscuous inhibitor.7
Uses
RK 682 is a protein tyrosine phosphatase 1B (PTP1B) inhibitor, a potential therapeutic strategy for curing insulin resistance.
References
1) Hamaguchi?et al. (1995),?RK-682, a potent inhibitor of tyrosine phosphatase, arrested the mammalian cell cycle progression at G1?phase; FEBS Lett.,?372?54 2) Shinagawa?et al. (1993),?Tetronic acid derivatives, its manufacturing methods and uses; Jpn.Kokai Tokkyo Koho JP 05-43568,?35?1791 3) Roggo?et al. (1994),?3-Alkanoyl-5-hydroxymethyl tetronic acid homologues and resistomycin; new inhibitors of HIV-1 protease; J. Antibiot (Tokyo),?47?136 4) Ishida?et al. (2004),?Structure-based design of a selective heparanase inhibitor as an antimetastatic agent; Mol. Cancer Ther.,?3?1069 5) Sodeoka?et al1. (1996),?Asymmetric synthesis of RK-682 and its analogs, and evaluation of their protein phosphatase inhibitory activities; Tet. Lett.,?37?8775 6) Sodeoka?et al. (2001),?Asymmetric Synthesis of a 3-Acyltetronic Acid Derivative, RK-682, and Formation of Its Calcium Salt during Silica Gel Chromatography; Chem. Pharm. Bull.,?49?206 7) Carneiro?et al. (2015),?Is RK-682 a promiscuous enzyme inhibitor? Synthesis and in vitro evaluation of protein tyrosine phosphatase inhibition of racemic RK-682 and analogues; Eur. J. Med. Chem.,?97?42
(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone Preparation Products And Raw materials
Raw materials
Preparation Products
(R)-4-Hydroxy-5-(hydroxymethyl)-3-(1-oxohexadecyl)-2(5H)-furanone Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Aladdin Scientific | +1-+1(833)-552-7181 | sales@aladdinsci.com | United States | 52927 | 58 |
| Energy Chemical | 021-58432009 400-005-6266 | marketing1@energy-chemical.com | China | 44843 | 58 |
| TargetMol Chemicals Inc. | 15002134094 | marketing@targetmol.cn | China | 28070 | 58 |
| Supplier | Advantage |
|---|---|
| Aladdin Scientific | 58 |
| Energy Chemical | 58 |
| TargetMol Chemicals Inc. | 58 |