Description
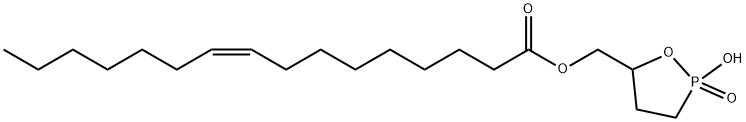
Cyclic phosphatidic acids (cPAs) are naturally occurring analogs of lysophosphatidic acid (LPA) in which the sn-2 hydroxy group forms a 5-membered ring with the sn-3 phosphate.1,2 Carba-derivatives of cPA (ccPA) are modified at the sn-2 (2-ccPA) or sn-3 (3-ccPA) linkage, preventing the opening of cPA to produce lysophosphatidic acid (LPA).3 Palmitoleoyl 3-carbacyclic phosphatidic acid (3-ccPA 16:1) is a cyclic LPA analog that contains the 16:1 fatty acid, palmitoleate, at the sn-1 position of the glycerol backbone.3 At 25 μM, it inhibits the transcellular migration of MM1 cells across mesothelial cell monolayers in response to fetal bovine serum (86.9%) or LPA (99.9%) without affecting proliferation.3 3-ccPA 16:1 significantly inhibits autotaxin (IC50 = 620 nM),4 an enzyme that is important in cancer cell survival, growth, migration, invasion and metastasis. When delivered intraperitoneally, 3-ccPA 16:1 significantly reduces the number of lung metastases formed in mice injected with B16F10 melanoma cells in the tail vein.4
References
1. Kobayashi, T., Tanaka-Ishii, R., Taguchi, R., et al. Existence of a bioactive lipid, cyclic phosphatidic acid, bound to human serum albumin Life Sci. 65(21),2185-2191(1999).
2. Mukai, M., Imamura, F., Ayaki, M., et al. Inhibition of tumor invasion and metastasis by a novel lysophosphatidic acid (cyclic LPA) Int. J. Cancer 81,918-922(1999).
3. Uchiyama, A., Mukai, M., Fujiwara, Y., et al. Inhibition of transcellular tumor cell migration and metastasis by novel carba-derivatives of cyclic phosphatidic acid Biochim. Biophys. Acta 1771,103-112(2007).
4. Baker, D.L., Fujiwara, Y., Pigg, K.R., et al. Carba analogs of cyclic phosphatidic acid are selective inhibitors of autotaxin and cancer cell invasion and metastasis J. Biol. Chem. 281(32),22786-22793(2006).