GSK2982772
- CAS No.
- 1622848-92-3
- Chemical Name:
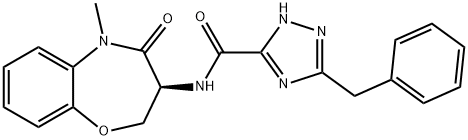
- GSK2982772
- Synonyms
- 5184;CS-2502;GSK2982772;GSK 2982772;GSK-2982772;RIP1 inhibitor GS2982772;(S)-5-BENZYL-N-(5-METHYL-4-OXO-2,3,4,5-TETRAHYDROBENZO[B][1,4]OXAZEPIN-3-YL)-4H-1,2,4-TRIAZOLE-3-CAR;5-benzyl-N-[(3S)-5-methyl-4-oxo-2,3,4,5-tetrahydro-1,5-benzoxazepin-3-yl]-1H-1,2,4-triazole-3-carboxamide;(S)-5-benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-4H-1,2,4-triazole-3-carboxamide;(S)-3-Benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-1H-1,2,4-triazole-5-carboxamide;1H-1,2,4-Triazole-5-carboxamide, 3-(phenylmethyl)-N-[(3S)-2,3,4,5-tetrahydro-5-methyl-4-oxo-1,5-benzoxazepin-3-yl]-
- CBNumber:
- CB53060417
- Molecular Formula:
- C20H19N5O3
- Molecular Weight:
- 377.4
- MOL File:
- 1622848-92-3.mol
- Modify Date:
- 2023/9/20 16:43:00
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H315-H319-H335 | |||||||||
| Precautionary statements | P261-P305+P351+P338 | |||||||||
| NFPA 704 |
|
GSK2982772 Chemical Properties,Uses,Production
Description
(S)-5-benzyl-N-(5-methyl-4-oxo-2,3,4,5-tetrahydrobenzo[b][1,4]oxazepin-3-yl)-4H-1,2,4-triazole-3-carboxamide, also known as GSK2982772, is a first-in-class receptor interacting protein 1(RIP1)-kinase specific clinical candidate for the treatment of inflammatory disease. It is an inhibitor of RIP1 that regulates necroptosis and inflammation and may play an important role in contributing to a variety of human pathologies, including immune-mediated inflammatory diseases. It has excellent activity in blocking many TNF-dependent cellular responses. Highlighting its potential as a novel anti-inflammatory agent, the inhibitor was also able to reduce spontaneous production of cytokines from human ulcerative colitis explants.
References
Harris, P. A., et al. "Discovery of a First-in-Class Receptor Interacting Protein 1 (RIP1) Kinase Specific Clinical Candidate (GSK2982772) for the Treatment of Inflammatory Diseases." Journal of Medicinal Chemistry 60.4(2017):1247-1261.
Jeong, Jae Uk, et al. "Synthesis of ( S )-3-amino-benzo[ b ][1,4]oxazepin-4-one via Mitsunobu and S N Ar reaction for a first-in-class RIP1 kinase inhibitor GSK2982772 in clinical trials." Tetrahedron Letters 58.23(2017):2306-2308.
GSK2982772 Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| A.J Chemicals | 91-9810153283 | New Delhi, India | 6124 | 58 | Inquiry |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | China | 32835 | 60 | Inquiry |
| career henan chemical co | +86-0371-86658258 +8613203830695 | China | 29899 | 58 | Inquiry |
| BOC Sciences | +1-631-485-4226 | United States | 19553 | 58 | Inquiry |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | China | 39916 | 58 | Inquiry |
| Zhejiang J&C Biological Technology Co.,Limited | +1-2135480471 +1-2135480471 | China | 10522 | 58 | Inquiry |
| changzhou huayang technology co., ltd | +8615250961469 | China | 9740 | 58 | Inquiry |
| InvivoChem | +1-708-310-1919 +1-13798911105 | United States | 6393 | 58 | Inquiry |
| Nanjing Doge Biomedical Technology Co., Ltd | +86-25-58227606 +86-15305155328 | China | 4128 | 58 | Inquiry |
| LEAP CHEM CO., LTD. | +86-852-30606658 | China | 24738 | 58 | Inquiry |
1622848-92-3(GSK2982772)Related Search:
1of4
chevron_right




