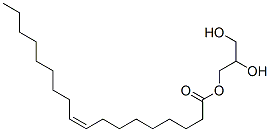
MONOOLEIN
- CAS No.
- 25496-72-4
- Chemical Name:
- MONOOLEIN
- Synonyms
- GLYCEROL MONOOLEATE;peceol;1-OLEOYL-RAC-GLYCEROL;1-MONOOLEIN;E 496;s1096;s1097;OP 85;SP 40;SP 65
- CBNumber:
- CB5757259
- Molecular Formula:
- C21H40O4
- Molecular Weight:
- 356.54
- MOL File:
- 25496-72-4.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/7/24 13:33:21
| Melting point | 35-37 °C |
|---|---|
| Boiling point | 449.35°C (rough estimate) |
| Density | 0.96 g/cm3 (25 ºC) |
| refractive index | 1.4626 (589.3 nm 40℃) |
| FEMA | 2526 | GLYCERYL MONOOLEATE |
| storage temp. | −20°C |
| solubility | chloroform: 50 mg/mL, clear, colorless |
| Water Solubility | <3.57mg/L(30 ºC) |
| Stability | Hygroscopic |
| CAS DataBase Reference | 25496-72-4(CAS DataBase Reference) |
| EPA Substance Registry System | Glyceryl monooleate (25496-72-4) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H319 |
| Precautionary statements | P264-P280-P305+P351+P338-P337+P313P |
| WGK Germany | 1 |
| F | 3 |
| HS Code | 3822000002 |
MONOOLEIN price More Price(3)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | PHR1484 | Glyceryl Monooleate Pharmaceutical Secondary Standard; Certified Reference Material | 25496-72-4 | 1G | ₹19950.48 | 2022-06-14 | Buy |
| ottokemi | G 1388 | Glycerol monooleate | 25496-72-4 | 100gm | ₹1809 | 2022-05-26 | Buy |
| ottokemi | G 1388 | Glycerol monooleate | 25496-72-4 | 500gm | ₹3105 | 2022-05-26 | Buy |
MONOOLEIN Chemical Properties,Uses,Production
Description
§ 184.1323(a) Glyceryl monooleate is prepared by esterification of commercial oleic acid that is derived either from edible sources or from tall oil fatty acids meeting the requirements of §172.862. It contains glyceryl monooleate (C21H40O4 and glyceryl esters of fatty acids present in commercial oleic acid.
Chemical Properties
Colourless Solid
Uses
93% 1-Oleoyl, 7% 2-oleoyl
Production Methods
Glyceryl monooleate is prepared by the esterification of glycerol with fatty acids, chiefly oleic acid. As the fatty acids are not pure substances, but rather a mixture of fatty acids, the product obtained from the esterification will contain a mixture of esters, including stearic and palmitic. Di- and triesters may also be present. The composition and, therefore, the physical properties of glyceryl monooleate may thus vary considerably from manufacturer to manufacturer; e.g. the melting point may vary from 10–35°C.
Definition
An antifog agent and dispersant.
Pharmaceutical Applications
Glyceryl monooleate is a polar lipid that swells in water to give
several phases with different rheological properties. It is available
in both nonemulsifying (n/e) and self-emulsifying (s/e) grades, the
self-emulsifying grade containing about 5% of an anionic surfactant.
The nonemulsifying grade is used in topical formulations as an
emollient and as an emulsifying agent for water-in-oil emulsions. It
is also a stabilizer for oil-in-water emulsions. The self-emulsifying
grade is used as a primary emulsifier for oil-in-water systems.
Glyceryl monooleate gels in excess water, forming a highly
ordered cubic phase that can be used to sustain the release of
various water-soluble drugs.It is also the basis of mucoadhesive
drug delivery systems.
Glyceryl monooleate is reported to enhance transdermal and
buccal penetration.
Safety Profile
When heated to decomposition it emits acrid smoke and irritating fumes.
Safety
Glyceryl monooleate is used in oral and topical pharmaceutical formulations and is generally regarded as a relatively nonirritant and nontoxic excipient.
storage
Glyceryl monooleate should be stored in an airtight container, protected from light in a cool, dry place.
Incompatibilities
Glyceryl monooleate is incompatible with strong oxidizing agents. The self-emulsifying grade is incompatible with cationic surfactants.
Regulatory Status
GRAS listed. Included in the FDA Inactive Ingredients Database (oral capsules, oral powder, oral tablets; creams, controlled-release transdermal films). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
MONOOLEIN Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ESTELLE CHEMICALS PVT LTD | +91-7620604545 +91-7620604545 | Maharashtra, India | 30 | 58 | Inquiry |
| VA SUDHA Chemicals Private Limited | +91-9653313056 +91-9653313056 | Maharashtra, India | 8 | 58 | Inquiry |
| HJ AROCHEM | +91-8690768060 +91-9724420086 | Ahmedabad, India | 171 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Mohini Organics | 8831043/53/56 | New Delhi, India | 55 | 50 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| Camex Speciality Private Limited | 08046051668 | Ahmedabad, India | 117 | 58 | Inquiry |
| Univenture Industries Pvt Ltd | 08048372658Ext 843 | Nagpur, India | 11 | 58 | Inquiry |
| Jeevika Spak Chemical | 08048250388Ext 872 | Nagpur, India | 4 | 58 | Inquiry |
| Devrk Enterprises | 08066085970Ext 164 | Madhya Pradesh, India | 2 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| ESTELLE CHEMICALS PVT LTD | 58 |
| VA SUDHA Chemicals Private Limited | 58 |
| HJ AROCHEM | 58 |
| CLEARSYNTH LABS LTD. | 58 |
| Mohini Organics | 50 |
| Otto Chemie Pvt. Ltd. | 58 |
| Camex Speciality Private Limited | 58 |
| Univenture Industries Pvt Ltd | 58 |
| Jeevika Spak Chemical | 58 |
| Devrk Enterprises | 58 |
25496-72-4(MONOOLEIN)Related Search:
1of4
chevron_right




