GADOLINIUM
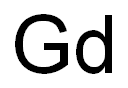
- CAS No.
- 7440-54-2
- Chemical Name:
- GADOLINIUM
- Synonyms
- Gd;GD007910;GD007925;GD005105;GD007905;GADOLINIUM;Gadolinium foil;gadolinium atom;GADOLINIUM METAL;Gadolinium chips
- CBNumber:
- CB7393251
- Molecular Formula:
- Gd
- Molecular Weight:
- 157.25
- MOL File:
- 7440-54-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/5/31 13:56:46
| Melting point | 1313 °C (lit.) |
|---|---|
| Boiling point | 3273 °C (lit.) |
| Density | 7.886 g/mL at 25 °C (lit.) |
| Flash point | 3266°C |
| solubility | soluble in dilute acid solutions |
| form | ingot |
| color | Silver |
| Specific Gravity | 7.898 |
| Resistivity | 126 μΩ-cm, 20°C |
| Water Solubility | Soluble in magnesium and diluted acid. Insoluble in water. |
| Sensitive | Air & Moisture Sensitive |
| Merck | 13,4347 |
| Exposure limits |
ACGIH: TWA 2 ppm; STEL 4 ppm OSHA: TWA 2 ppm(5 mg/m3) NIOSH: IDLH 25 ppm; TWA 2 ppm(5 mg/m3); STEL 4 ppm(10 mg/m3) |
| Stability | Stable. Powder is highly flammable. Incompatible with strong oxiziding agents, halogens, acids. Powder may react with water or moisture. |
| CAS DataBase Reference | 7440-54-2(CAS DataBase Reference) |
| EPA Substance Registry System | Gadolinium (7440-54-2) |
| Poissons Ratio | 0.259 |
|---|---|
| Shear Modulus | 21.8 GPa, Calculated |
| Hardness, Vickers | 37, polycrystalline |
| Bulk Modulus | 37.9 GPa |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS02 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H228-H261 | |||||||||
| Precautionary statements | P210-P231+P232-P240-P241-P280-P370+P378 | |||||||||
| Hazard Codes | F,Xi | |||||||||
| Risk Statements | 15-36/38 | |||||||||
| Safety Statements | 43-7/8-26-Neverusewater. | |||||||||
| RIDADR | UN 3208 4.3/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | LW3850000 | |||||||||
| F | 1 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 2846902015 | |||||||||
| NFPA 704 |
|
GADOLINIUM price More Price(34)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | 693723 | Gadolinium foil, thickness 1?mm, 99.9% trace rare earth metals basis | 7440-54-2 | 1EA | ₹20762.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 263087 | Gadolinium chips | 7440-54-2 | 10G | ₹13113.65 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 261114 | Gadolinium ingot, 99.9% trace rare earth metals basis | 7440-54-2 | 10G | ₹8640.45 | 2022-06-14 | Buy |
| ALFA India | ALF-045929-KS | Gadolinium sputtering target, 76.2mm dia x 3.18mm thick | 7440-54-2 | 1ea. | ₹126975 | 2022-05-26 | Buy |
| ALFA India | ALF-045915-14 | Gadolinium powder, -200 mesh, 99.9% (REO) | 7440-54-2 | 25g | ₹50974 | 2022-05-26 | Buy |
GADOLINIUM Chemical Properties,Uses,Production
Chemical Properties
metal foil, chunks or powder. The powder of gadolinium is highly flammable; incompatible with strong oxidising agents, halogens, acids; and reacts with water or moisture.
Physical properties
Gadolinium is silvery-white, soft, malleable, and ductile with a metallic luster. It is the secondof what is referred to as the dysprosium, subgroup in the middle of the lanthanide seriesof rare-earths. It tarnishes in air, forming the oxide (Gd2O3) on the surface, which flakes offthe surface, exposing a fresh metal that in turn oxidizes.
Its melting point is 1,313°C, its boiling point is 3,273°C, and its density is 7.90g/cm3.
Isotopes
There are 39 isotopes of gadolinium. Seven of these are stable. They are: Gd-54, which makes up 2.18% of all the gadolinium found in the Earth’s crust; Gd-55,supplying 14.80%; Gd-156, making up 20.47%; Gd-157, constituting 15.56%; and Gd-158, accounting for 24.85%. In addition, there are two isotopes of gadolinium that areradioactive and with such long half-lives that they still exist in the Earth’s crust. They areregarded as stable isotopes along with the other seven. They are Gd-152 (1.08×10+14years), which exists in just 0.20% in abundance, and Gd-160 (1.3×10+21 years), foundin 21.86% abundance.
Origin of Name
Named for the mineral gadolinite, which was named for the French chemist Johann Gadolin.
Occurrence
Gadolinium is the 40th most abundant element on Earth and the sixth most abundant ofthe rare-earths found in the Earth’s crust (6.4 ppm). Like many other rare-earths, gadoliniumis found in monazite river sand in India and Brazil and the beach sand of Florida as well asin bastnasite ores in southern California. Similar to other rare-earths, gadolinium is recoveredfrom its minerals by the ion-exchange process. It is also produced by nuclear fission in atomicreactors designed to produce electricity.
Characteristics
Gadolinium, unlike most of the rare earths in the dysprosium subgroup, reacts slowlywith water, releasing hydrogen. It is strongly magnetic at low temperatures. Two of its stableisotopes (Gd-155 and Gd-157) have the greatest ability of all natural elements to absorb thermalneutrons to control the fission chain reaction in nuclear reactors. However, few of theseisotopes are found in the ores of gadolinium.
Uses
Gadolinium’s main use is based on its ability to absorb neutrons, thus making it ideal as aneutron-shielding and neutron-absorbing metal. It is also used as an alloying agent for steel andother metals to make the metals more workable and to be able to withstand low temperatures.
Gadolinium is used in the manufacture of electronics and can be combined with yttriumto make garnets used in microwaves. Gadolinium is used as a catalyst to speed up chemicalreactions, and to activate phosphor compounds in TV screens and cast filaments in electricaldevices. It is also used in high-temperature furnaces. Gadolinium is paramagnetic at normalroom temperatures (weaker than ferromagnetic) and becomes strongly ferromagnetic at verycold temperatures.
Definition
A ductile malleable silvery element of the lanthanoid series of metals. It occurs in association with other lanthanoids. Gadolinium is used in alloys, magnets, and in the electronics industry. Symbol: Gd; m.p. 1313°C; b.p. 3266°C; r.d. 7.9 (25°C); p.n. 64; r.a.m. 157.25.
Hazard
The halogens of gadolinium are very toxic, and gadolinium nitrate is explosive. As withmost rare-earths, care should be taken not to inhale fumes or ingest particles of gadolinium.
GADOLINIUM Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Aritech Chemazone Private Limited | +91-9034345475 | Punjab, India | 684 | 58 | Inquiry |
| Central Drug House(P) Ltd. | 91-11-49404040 | New Delhi, India | 6160 | 58 | Inquiry |
| Parmanu Dhatu Nigam | 08048372587Ext 498 | Mumbai, India | 27 | 58 | Inquiry |
| Harsh Trading Company | 08048372590Ext 538 | Mumbai, India | 4 | 58 | Inquiry |
| Intelligent Materials Private Limited | 08048953178 | Punjab, India | 197 | 58 | Inquiry |
| Nano Research Elements | 08048372588Ext 365 | Delhi, India | 101 | 58 | Inquiry |
| Rare Earth (India) Unit Of Vijay Commercial House | 08046072837 | Mumbai, India | 32 | 58 | Inquiry |
| Otto Chemie Pvt. Ltd. | +91 9820041841 | Mumbai, India | 5873 | 58 | Inquiry |
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Alfa Aesar | 1 800 209 7001 | Maharashtra, India | 6913 | 58 | Inquiry |
Related articles
- Gadolinium:Discovery,Minerals,Uses,Safety
- Gadolinium is a silvery-white metal when oxidation is eliminated. It is only slightly malleable and is a ductile REE. Gadolini....
- May 31,2024
7440-54-2(GADOLINIUM)Related Search:
1of4
chevron_right




