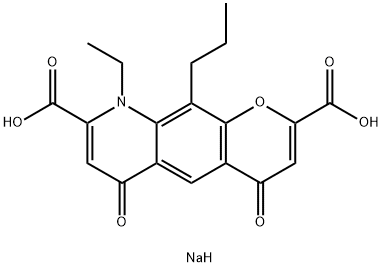
NEDOCROMIL SODIUM
- CAS No.
- 69049-74-7
- Chemical Name:
- NEDOCROMIL SODIUM
- Synonyms
- Tayled;Tilade;Alocril;Tilarin;Unii-et8if4ks1t;Nedocromil sodium;NEDOCROMIL DISODIUM SALT;NEDOCROMIL SODIUM###Nedocromil;Sodium 9-ethyl-4,6-dioxo-10-propyl-6,9-dihydro-4H-pyrano[3,2-g]quinoline-2,8-dicarboxylate;4H-Pyrano[3,2-g]quinoline-2,8-dicarboxylicacid, 9-ethyl-6,9-dihydro-4,6-dioxo-10-propyl-, sodium salt
- CBNumber:
- CB81011490
- Molecular Formula:
- C19H18NNaO7
- Molecular Weight:
- 395.34
- MOL File:
- 69049-74-7.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/6/8 9:02:09
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H319 | |||||||||
| Precautionary statements | P264-P280-P305+P351+P338-P337+P313P | |||||||||
| WGK Germany | 3 | |||||||||
| NFPA 704 |
|
NEDOCROMIL SODIUM price More Price(2)
NEDOCROMIL SODIUM Chemical Properties,Uses,Production
Description
Nedocromil sodium is an antiallergic agent with broader mast cell stabilizing properties than related cromolyn sodium. Administered as an aerosol, it is useful in the treatment of bronchial asthma and related diseases.
Uses
Nedocromil Sodium (Technical Grade) is a medication considered as mast cell stabilizer which act to prevent wheezing, shortness of breath, and other breathing problems caused by asthma.
Indications
nedocromil sodium(Tilade) is chemically related drugs called chromones that is used for the prophylaxis of mild or moderate asthma. It is administered by inhalation and has very good safety profiles, making them particularly useful in treating children.
General Description
Nedocromil sodium, disodium9-ethyl-6, 9-dihydro-4,6-dioxo-10-propyl-4H-pyrano[3, 2-g]quinoline-2,8-dicarboxylate (Tilade), is structurally relatedto cromolyn and displays similar, but broader, pharmacologicalactions. It was available as an aerosol in ametered-dose inhaler for asthma treatment, and currently remainsavailable as ophthalmic solution for the treatmentof itching associated with allergic conjunctivitis. The inhalationformulation was marketed for maintenance therapyin the management of patients with mild-to-moderatebronchial asthma, but since has been withdrawn.
Pharmacokinetics
Nedocromil sodium was developed by changing the furan ring of khellin to a piperidinone ring. In vitro, nedocromil sodium inhibits the release of inflammatory response mediators from a variety of cells, including neutrophils, mast cells, macrophages, and platelets. Inhaled nedocromil sodium is poorly absorbed into the systemic circulation, with approximately 3% of an inhaled dose excreted in the urine during the first 6 hours after administration. Only 2% of orally dosed nedocromil sodium is bioavailable, 89% of which is protein bound. When administered IV, nedocromil sodium is not metabolized and is excreted unchanged in the bile and the urine. Nedocromil sodium is available in aerosol canisters for oral inhalation via a mouthpiece.
Clinical Use
nedocromil sodium is used almost exclusively for the prophylactic treatment of mild to moderate asthma and should not be used for the control of acute bronchospasm. This agent is effective in about 60 to 70% of children and adolescents with asthma. Unfortunately, there is no reliable means to predict which patients will respond.It is less effective in older patients and in patients with severe asthma. It may take up to 4 to 6 weeks of treatment for cromolyn sodium to be effective in chronic asthma, but it is effective after a single dose in exercise-induced asthma. With respect to clinical efficacy, cromolyn sodium and nedocromil sodium do not differ in a substantial way.
Side effects
nedocromil sodium is the least toxic of available therapies for asthma. Adverse reactions are rare and generally minor. Those occurring in fewer than 1 in 10,000 patients include transient bronchospasm, cough or wheezing, dryness of throat, laryngeal edema, swollen parotid gland, angioedema, joint swelling and pain, dizziness, dysuria, nausea, headache, nasal congestion, rash, and urticaria.
NEDOCROMIL SODIUM Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| BOC Sciences | +1-631-485-4226 | United States | 19553 | 58 | Inquiry |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | China | 49391 | 58 | Inquiry |
| Hebei Bonster Technology Co.,Limited | +8613315996897 | China | 796 | 58 | Inquiry |
| TargetMol Chemicals Inc. | +1-781-999-5354 +1-00000000000 | United States | 19892 | 58 | Inquiry |
| AFINE CHEMICALS LIMITED | +86-0571-85134551 | China | 15395 | 58 | Inquiry |
| Dayang Chem (Hangzhou) Co.,Ltd. | 571-88938639 +8617705817739 | China | 52861 | 58 | Inquiry |
| LEAP CHEM CO., LTD. | +86-852-30606658 | China | 24738 | 58 | Inquiry |
| GIHI CHEMICALS CO.,LIMITED | +8618058761490 | China | 49999 | 58 | Inquiry |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 | China | 33350 | 58 | Inquiry |
69049-74-7(NEDOCROMIL SODIUM)Related Search:
1of4
chevron_right




